The colloquium of the Department of Chemical and Biological Engineering takes place at 16.15 on many Thursdays, usually during the lecture period, in the Hanns-Hofmann Hörsaal (KS I, Cauerstr. 4). The talks are given by invited national and international researchers and cover critical themes in current research. The lectures, given in English, are targeted to a broad interdisciplinary audience so that students and researchers of the department and beyond can follow. The colloquia are of particular benefit for Master’s and Bachelor’s students in order to gain insight into the wide variety of subfields in chemical and biological engineering and assist with their choice of future project topics.
Prof. Dr. Aljoscha Wahl
Konrad-Zuse-Str. 3
91052 Erlangen
Deutschland
- Telefon: +49913185-23005
- E-Mail: aljoscha.wahl@fau.de
- Webseite: https://www.bvt.tf.fau.de/
Unless stated all talks at 16.15 in the Hanns-Hofmann-Hörsaal (KS I, Cauerstr. 4).
Winter term 2025/26

Title: Understanding Physical Properties in the Context of CCUS and H2 Projects
The technologies of carbon capture and hydrogen production, along with transportation and storage processes, are set to contribute significantly in the ongoing energy transition. The two principle actors in these processes, CO2 and H2, appear in combination with myriad other substances in complex combinations, the understanding of which is crucial to process design and operation. Physical properties of mixtures containing CO2 and/or H2 are crucial in this context. Phase behaviour and phase properties must be known under a wide range of operating conditions including both low and high temperatures and often also high pressures. This present significant challenges, for example in setting and meeting CO2 purity specifications for pipeline and shipping operations, in ensuring reliable fiscal metering of hydrogen, and in the quest for improved carbon capture solvents. The lecture will explore some of the physical property challenges currently facing the CCUS and H2 industries and present some of the research currently being carried out to address them.
CV
2001-date: Professor of Thermophysics, Chemical Engineering Department, Imperial College London
1995-2001: Senior Lecturer, Chemical Engineering Department, Imperial College London
1988-1995: Lecturer, Chemical Engineering Department, Imperial College London
1985-1987: Ramsay Memorial Fellow, Chemistry Department, University College London
1984-1985: Lindemann Trust Fellow, National Institute of Standards & Technology, USA
1980-1984: PhD in Physical Chemistry, University College London, Thesis title: “The speed of sound in gases”
1977-1980: BSc with 1st class honours in Chemistry, University College London

Title: From Ice to Pores: Shaping Drug Stability through Freezing and Annealing
Abstract:
Freeze-drying is widely used to stabilise modern medicines, but the process is slow, energy‑intensive and still relies heavily on trial and error in development. In this talk, a simple sugar–water system is used to demonstrate how the freezing process and subsequent low-temperature treatment determine the invisible pore network through which ice later escapes as vapour. Combining in‑situ microscopy experiments with tailored computer simulations, the work tracks how ice crystals grow, merge and rearrange during freezing and annealing, and how this reshapes the porous structure. The results provide practical rules for linking process conditions to pore size and connectivity, and demonstrate how digital models can complement experiments to design more robust, efficient freeze‑drying cycles.
CV:
Dr. Sudeshna Roy is Privatdozentin at Friedrich-Alexander-Universität Erlangen–Nürnberg (FAU) and Manager for Process Simulation at Daiichi Sankyo Europe GmbH. She received her PhD from the University of Twente in 2018 and completed her habilitation at FAU in 2025. After her PhD, she held postdoctoral positions at Pennsylvania State University, Syracuse University (USA), and Heriot-Watt University (UK). Her research focuses on granular materials, particle-based simulation methods, and pharmaceutical process modeling, particularly freeze-drying and formulation processes. She has authored multiple peer-reviewed publications and collaborates closely with both academia and industry.
Summer Semester 2025

Title: Multi-enzyme cascade reactions for a renewable biotechnology
– from biobased polymers to photobiocatalysis
Cascade and one pot reactions represent an exciting and recent development in biocatalysis. There are, however, a range of technological and scientific challenges to be met en route to industrial scale. In this talk, I will focus on the coupling of biocatalysts to other catalysts in order to provide routes that would be difficult with the individual, separated reactions.
A first example presents the synthesis of biobased olefins as highly promising polymer precursors. The alpha-methylene lactone Tulipalin A has two polymerizable functional moieties and is a potential substitute of (meth)acrylates in vinyl-addition polymerization and (co)monomer for lactone ring-opening polymerization. While Tulipalin A can be isolated from, its biosynthesis remains unknown. A possible synthesis route starts from the microbial terpenoid metabolism. The key step of this pathway is catalyzed by a membrane-bound monooxygenase. Rational design of the activity of this this membrane-enzyme aiming based on de novo structure predictions will be presented.
A second example deals with the coupling of enzymes with photosynthetic water-splitting as means to provide electrons and oxygen for redox biotransformation. This approach greatly improves the atom economy of redox biotransformations and circumvents problems associated to the low mass-transfer of oxygen. Several examples demonstrated the feasibility of the approach with high selectivity and high reaction rates for several redox processes. The presentation will discuss strategies how to overcome current limitations and to improve the volumetric productivity by combining process design and metabolic engineering, and present a strategy towards clean redox biocatalysis in engineered autotrophic microorganisms.
CV Prof. Dr. Robert Kourist
Dr. Robert Kourist is full Professor of Molecular Biotechnology and head of the institute with the same name at Graz University of Technology, Graz, Austria and Leader of the Area Biotransformation at the Austrian Centre of Industrial Biotechnology. He received his diploma in biochemistry from the University of Greifswald, Germany, in 2006. After obtaining his PhD in 2008 under the supervision of Prof. Uwe Bornscheuer at the University of Greifswald, he went for one year as postdoctoral fellow to the laboratory of Prof. Kenji Miyamoto at the Keio University in Yokohama, Japan. In 2009, he received the DSM Science and Technology Award North and a 1st prize in the VentureCup Mecklenburg-Vorpommern. In 2015, he was appointed member of the Young College of the Academy of Sciences and Arts of Northrhine Westphalia. From 2012 to 2016, he was Junior Professor for Microbial Biotechnology at the Ruhr-University Bochum. Dr. Kourist’s expertise lies in the field of biocatalysis with a special focus on the development of chemo-enzymatic cascade reactions and light-catalyzed redox reactions. He has co-authored more than 120 publications and patents and has an H-index of 37.

Title: Non-Linear Super-Stencils for Turbulence Model Corrections
Accurate simulation of turbulent flows remains a challenge due to the high computational cost of direct numerical simulations (DNS) and the limitations of traditional turbulence models. This research explores a novel approach to augmenting standard models for Reynolds-Averaged Navier-Stokes (RANS) simulations using a Non-Linear Super-Stencil (NLSS). The proposed method introduces a fully connected neural network that learns a mapping from the local mean flow field to a corrective force term, which is added to a standard RANS solver in order to align its solution with high-fidelity data; an illustration of an NLSS is shown in Fig. 1. 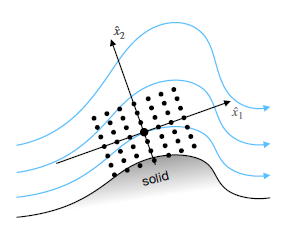
Figure 1: Non-Linear Super-Stencil (black dots): The stencil is centered around the point at which the correction force shall be determined, and it is aligned with the mean velocity at its center. A procedure is devised to extract training data from reference DNS and large eddy simulations (LES). To reduce the complexity of the non-linear mapping, the dimensionless local flow data is aligned with the local mean velocity, and the local support domain is scaled by the turbulent integral length scale. After being trained on a single periodic hill case, the NLSS-corrected RANS solver is shown to generalize to different periodic hill geometries and different Reynolds numbers, producing significantly more accurate solutions than the uncorrected RANS simulations. For demonstration, Fig. 2 shows RANS simulation results with and without NLSS-correction along with high fidelity references (mean velocity profiles in the top plots and wall shear stresses on the bottom) of two test cases which were not used for training. For training a domain of lenth L = 9m, a hill stretch factor of alpha = 1 and a Reynolds number of Re = 10595 were considered, while L = 13:929m, alpha = 1:5 and Re = 5600 were chosen for the left test case and L = 9m, alpha = 1 and Re = 19000 for right test case. For both test cases it can be observed that the NLSS-corrected RANS solutions closely agree with the reference data (much better than the uncorrected ones), thus demonstrating the ability of the NLSS to generalize across different geometries and Reynolds numbers. 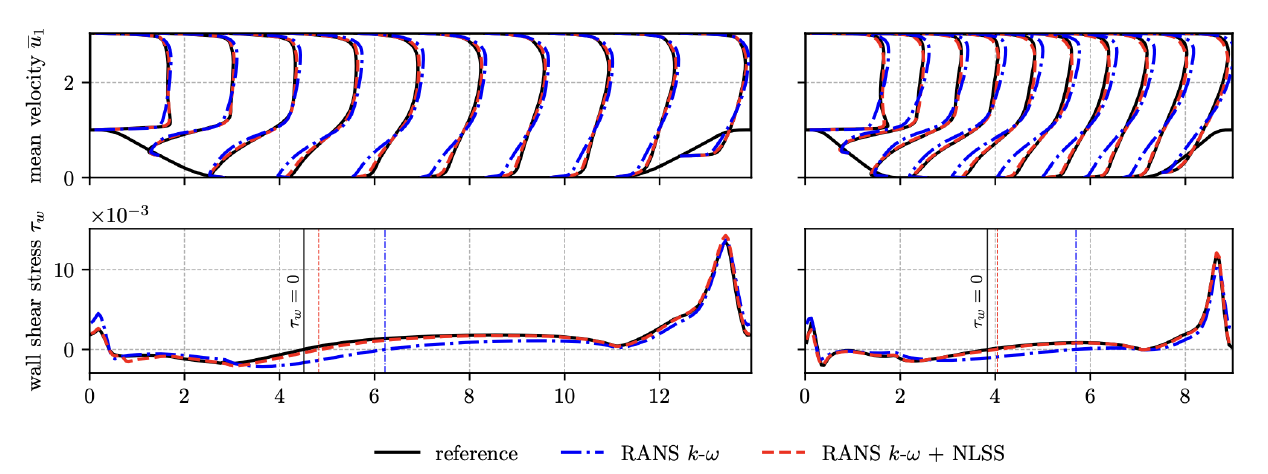
Figure 2: Generalization: RANS simulation results of two test cases (which were not used for training) with and without NLSS-correction along with high fidelity data. Shown on the top are profiles of the horizontal mean velocity component at ten different downstream locations, and on the bottom the wall shear stresses along the bottom wall of the periodic hill geometries. Locations of mean flow re-attachment of reference and NLSS-corrected solutions are marked by the vertical lines. While the ability of NLSS for model correction is demonstrated here for turbulent flows, the concept is more general and will be explored in the future for other systems of partial differential equations
involving physical models.

Title: Model-assisted Analysis and Design of Electrochemical Technologies
Electrochemical technologies offer efficient and dynamic storage of electrical energy in batteries or – via electrolysis – in hydrogen. In addition, power-to-chemicals technologies, such as electrochemical CO2 conversion or production of expensive fine chemicals, promise a deep energy transition of the chemical industry. Almost all new electrochemical technologies, but also established ones, suffer from performance losses due to a lack of quantitative insight into processes in the cells. Modelling tools at higher levels are already well established and predictive, when it comes to heat and mass transfer, as these phenomena are widespread in (chemical) engineering. They struggle, however, to reproduce or predict the electrochemical behaviour of electrodes.
This talk covers established and upcoming electrochemical energy and power-to-chemical technologies, their application and state of development. The complexity and challenges of the technologies at electrode level are complemented by strategies how to formulate and parameterise suitable models. The models are then used to give insight into the many non-measurable processes or states, and for virtual design of better catalysts, electrodes, cells or operating conditions.
CV
Ulrike Krewer Dr.-Ing. is a Full Professor and Head of the Institute for Applied Materials – Electrochemical Technologies at Karlsruhe Institute of Technology (KIT), Germany. She received her Dipl.-Ing. at FAU Erlangen-Nuremberg and a PhD in Process Engineering from University/Max Planck Institute Magdeburg. After researching in Samsung (Korea) and an Assistant Professorship, she became Full Professor for Energy and Process Systems Engineering at TU Braunschweig in 2012, and moved to her current position KIT in 2020. With more than 20 years of research expertise in electrochemical energy technologies, she and her team contribute(d) to advancing established technologies, such as Li-ion batteries and PEM electrolysis, as well as exploratory ones such as Li/Na-metal batteries and CO2 electrolysis. Model-based and dynamic analysis of processes at electrodes allows her to reveal performance limiting steps and the (degradation) state of cells and electrodes, and to propose improvements.
Winter Semester 2024/25
Prof. Dr. Johan Padding

Prof. Johan Padding is full professor and chair of Complex Fluid Processing in the Department of Process and Energy (P&E) of Delft University of Technology. He has an MSc in applied physics and a PhD in chemical physics from University of Twente, focusing on microscale and mesoscale simulations of soft matter. After several postdocs in Cambridge (UK), Twente (NL) and Louvain-la-Neuve (BE), Johan got a tenured position at Eindhoven University of Technology in 2011 where he shifted his attention to larger scale multiphase flows. He moved to Delft University of Technology in 2016. Currently, Johan does research on multiphase flows, mesoscale transport phenomena and heterogeneous catalysis, with applications in the fields of chemical conversions in fluidised beds, spray drying, crystallisation, and chemical reactor design, including electrochemical conversion.
Title: Modelling multiphase flow equipment for a sustainable future
The energy transition requires development of new conversion and separation equipment. The design and scale-up of such equipment requires fast models that can predict hydrodynamics, heat- and mass transfer, chemical conversion and phase transitions, usually in multi-phase systems. Unfortunately, these phenomena depend sensitively on small-scale events such as surface reactions or collisions between particles or droplets. It is therefore necessary to employ a multiscale modelling approach in which correlations are extracted from detailed small-scale models and fed into larger-scale models. In this talk I will give some examples from our group, highlighting our work on fluidised beds containing elongated particles and on electrochemical reactors for CO2 conversion. 
Professor Ioannis G. Economou is Visiting Professor in the Department of Chemical and Biological Engineering of Friedrich-Alexander-Universität Erlangen-Nürnberg. He holds a Diploma in Chemical Engineering from the National Technical University of Athens, Greece (1987) and a PhD also in Chemical Engineering from The Johns Hopkins University in Baltimore, Maryland, USA (1992). He was a post-doctoral researcher in Delft University of Technology in the Netherlands (1993 – 94) and in Exxon Research and Engineering Company, in New Jersey, USA (1994 – 95). From 1995 to 2009, he worked at the National Center for Scientific Research “Demokritos” in Athens, Greece where he held the position of Director of Molecular Thermodynamics and Modeling of Materials Laboratory from 2003 to 2021 (while on leave from 2009 to 2015, and 2016 to 2021). From 2009 until 2012, he was the Associate Provost for Graduate Studies and Professor of Chemical Engineering at the Petroleum Institute, Abu Dhabi. In 2013, he was appointed Professor of Chemical Engineering at Texas A&M University at Qatar (TAMUQ), in 2017 Associate Dean for Academic Affairs, in 2021 Senior Associate Dean for Academic Affairs and Graduate Studies and from July 2023 until July 2024, he was the Executive Associate Dean of TAMUQ. He is a Fellow of the American Institute of Chemical Engineers (2021).
He held various visiting / research positions including research fellow in University College London (1994 – 96 as a Marie Curie Fellow of the European Commission) and Princeton University (2004 and 2015), and visiting Professor in the Technical University of Denmark (2001 and 2006 – 07) and the American College of Greece (2007 – 09). Furthermore, he has consulted extensively for major oil and chemical companies in North America, Europe and Middle East.
Prof. Economou’s research interests are related to the development and validation of molecular and macroscopic thermodynamic models for the oil & gas, chemical and pharmaceutical industry. In recent years, he developed models for CO2 capture, transportation and sequestration technologies, shale gas technology, aqueous systems, green solvents, pharmaceuticals, and soft materials including organic and inorganic membranes, polymers, ionic liquids, metal organic frameworks, etc. He has been the project director for 30 research projects funded by the European Commission, national funding agencies in Greece and in Qatar, and industry, and principal investigator for another 17 projects. He has raised more than 10 million € of external research funding.
Prof. Economou has supervised 20 MSc students, 15 PhD students and 23 post-docs, he has published 240 peer-reviewed research papers in leading journals in Chemical Engineering, Physical Chemistry and Polymer Science. In addition, he co-authored 10 book chapters and co-edited 1 book entitled “Natural Gas Processing from Midstream to Downstream” (Wiley, 2019). His H-index is 59 according to Scholar Google. He has given approximately 400 presentations in conferences, Universities and industrial research centers worldwide. He has served as external examiner for 25 PhD theses in 11 countries (Europe, Latin America and Middle East). He is Editor of Fluid Phase Equilibria (Elsevier).
Title: From atoms and molecules to materials: Design of novel membranes for gas separation
The unprecedented increase of computing power at affordable price and the development of advanced computational techniques spanning from the sub-molecular all the way to the macroscopic engineering level has made computational molecular engineering a very powerful tool for the design of novel advanced materials with tailor-made properties. In this lecture, we will present a predictive model for the accurate design of highly selective Zeolitic-imidazolate Frameworks (ZIFs) for the separation of various gas mixtures, using a combination of Density Functional Theory (DFT) calculations, molecular simulation (both Molecular Dynamics and Grand Canonical Monte Carlo) and finally a Machine Learning model that allows massive screening of various structures whose relevant properties are generated by molecular simulation or experimental data [1]. Furthermore, a coarse-grained model based on Martini 3 representation will be shown to provide accurate structural and permeability predictions for various gases in ZIFs [2]. This approach can be generalized and used for other nanoporous materials for various applications. An immediate example refers to mixed matrix membranes where ZIFs are dispersed in a polymer matrix. In this application, a coarse-grained model for the polymer based on Martini 3 will be also presented [3]. Model predictions are in excellent agreement with experimental data [4].
References
[1] P. Krokidas, S. Karozis, S. Moncho, G. Giannakopoulos, E.N. Brothers, M.E. Kainourgiakis, I.G. Economou, T.A. Steriotis, J. Mater. Chem. A, 10, 13697-13703 (2022).
[2] A.M.O. Mohamed, I.G. Economou, H.-K. Jeong, J. Chem. Phys., 160, 204706 (2024).
[3] A.M.O. Mohamed, I.G. Economou, H.-K. Jeong, under review (2024).
[4] Y. Hua, A.M.O. Mohamed, G.M. Choi, K.Y. Cho, I.G. Economou, H-K. Jeong, ACS Appl. Mater. Interfaces, 16, 15273-15285 (2024).
Prof. Dr. Jörg Schumacher

Title: Boundary layer analysis in high-Rayleigh number turbulent convection
Turbulent convection processes are ubiquituous in nature and technology. We study the structure of the thermal and viscous boundary layers in three-dimensional Rayleigh-Bénard convection, a paradigm of these flows, for a range of Rayleigh numbers Ra=1e+5 to 1e+11 at Prandtl numbes of Pr=0.7 in direct numerical simulations. The convection flow evolves in a plane layer of aspect ratio 4H:4H:H with H being the layer height. We report the following findings: For the highest Rayleigh numbers, a hierarchy of thermal plumes is observed. Finer plumes close to the wall merge to bigger ones further away from the wall. The plume spacing is found to be self-similar, which suggests local boundary layer instability mechanisms at work. The velocity field close to the wall can be decomposed into shear-dominated (with different directions) and shear-free regions. There is no coherent shear flow covering the whole boundary plate, as assumed in several theories of turbulent heat transfer. We discuss possible connections of these findings to the scaling of the turbulent heat transfer with Rayleigh number which is close to a 1/3-power law.

Colloquia in Previous Semesters
Dr. Günter K. Auernhammer

The influence of complex rheology on the spreading of drops
Understanding the drop spreading behaviour of rheologically complex liquids, like dense granular suspensions or concentrated polymer solutions, is of crucial importance for applications such as inkjet printing, spraying paints or coating. In this presentation, we focus in the correlation between complex rheological behavior, like shear thickening or shear thinning, and the dynamics of drop spreading on hard substrates.
As shear thickening systems, we study the spreading behaviour of suspensions on clean hydrophilic glass surfaces, with increasing particle mass fractions (Φ = 0.3 to 0.65) and varying particle diameter (d = 10 to 20 μm), chemistry and morphology. Depending on the particle mass fraction, being close or not to the jamming transition of the suspension, two outcomes were observed: the spreading curve either exhibited a spreading behaviour qualitatively similar to the one of the Newtonian carrier liquid, with two spreading regimes or a spreading behaviour diverging from the reference, with three spreading regimes and an increase in spreading rate in-between two, see Fig. 1. The different spreading behavior strongly correlates with an increasing shear thickening of the suspensions. The correlation between rheological behavior and spreading dynamics is general for all investigates particle types.
The study is complemented by spreading experiments of shear thinning concentrated polymer solutions. These polymer solutions exhibit also strong correlation of the spreading dynamics with the rheological properties of the solutions, like the shear rate at the end of the Newtonian plateau or the exponent in an assumed power law model.
In both cases, we relate the observed correlations between rheological behaviour and spreading dynamics of drops to the dissipative mechanisms close to the moving contact line of the drop.
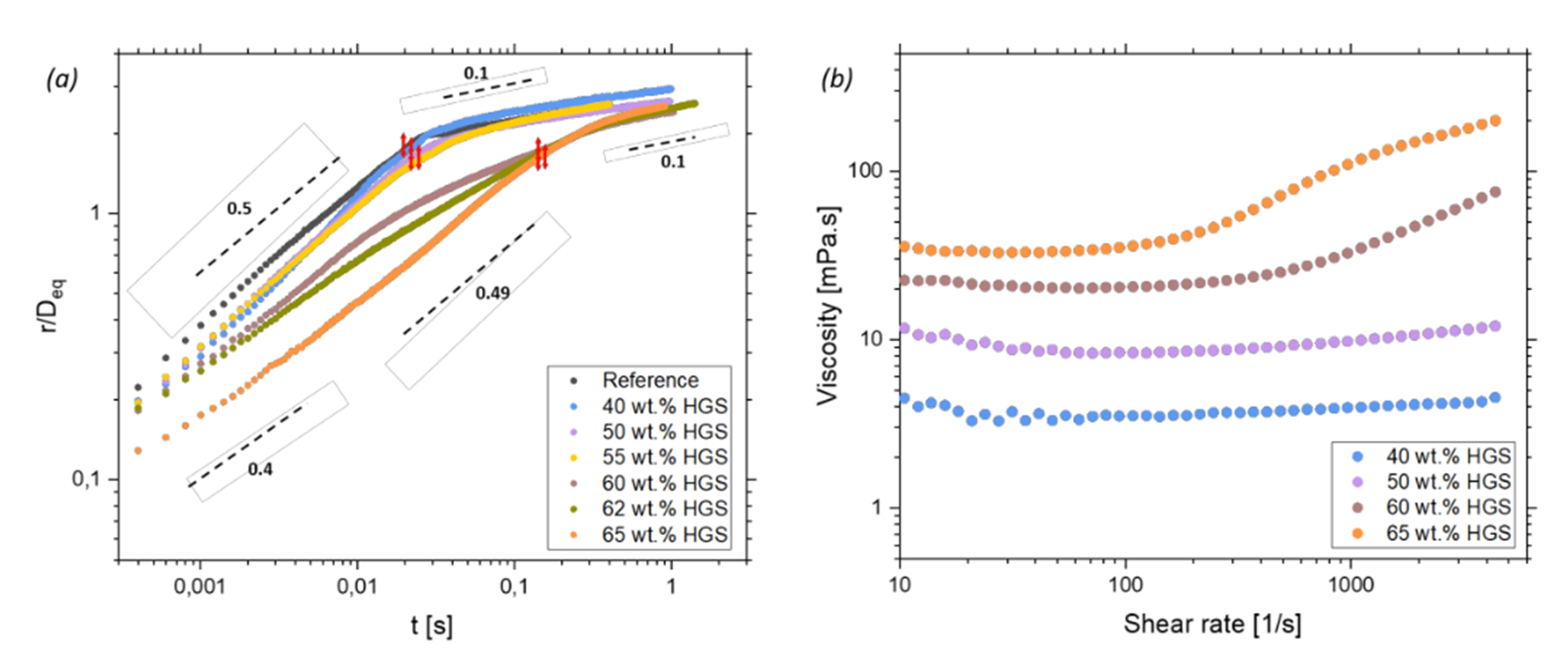
Figure 1: (a) Time evolution of the spreading radius for drops of aqueous suspensions with increasing particle mass fractions, (b) Flow curves of selected wt.% same suspensions.
tbd
Prof. Dr. Jürgen Dahlhaus

Nachhaltigkeit in der chemischen Industrie und bei der BASF
Die chemische Industrie steht vor großen Herausforderungen: Wir sind mitten in einer Trans-formation hin zu nachhaltigeren Produkten und Produktzyklen, deren Herstellung weniger Ressourcen verbraucht und weniger Emissionen erzeugt. Der Vortrag beschreibt, was das für eine Firma wie BASF bedeutet und wie wir diese Herausforderungen angehen. Es werden verschiedene Ansätze und Projekte vorgestellt und aufgezeigt, welche technischen Lösungen dafür notwendig sind. Abschließend wird kurz diskutiert, welche Beiträge die Hochschulen leisten können und was zukünftige Ingenieurinnen und Ingenieure erwartet.

4 May 2023 – Dr.-Ing. Sebastian Werner, Previously: Dataiku SAS, from June 2023: Thoughtworks Deutschland GmbH

Dr.-Ing. Sebastian Werner
Previously: Dataiku SAS
From June 2023: Thoughtworks Deutschland GmbH
LinkedIn Profile
Digitalization and AI in Chemical Engineering – Why we shouldn’t ignore recent developments like ChatGPT
Digitalization has become one of the most prominent topics for chemical businesses, impacting and transforming the product lifecycle, from research and development to production and operations.
One area where digital technologies are having a significant impact is through the use of machine learning and AI. It enables chemical engineers to optimize processes, predict outcomes, and generate insights that were previously impossible to obtain. By leveraging large amounts of data and applying advanced algorithms and machine learning techniques, AI is helping chemical engineers improve efficiency, reduce waste, and enhance safety.
 Seemingly unrelatedly, progress in fields like large-language models (e.g. ChatGPT) or generative AI (e.g. DALL-E or Google Parti) have made the headlines. But what does that mean for us? Generative AI, on the other hand cannot just produce pretty pictures as show here – it has the potential to transform the way chemical engineers approach the design of new materials, chemicals and even suggest process flowsheets or engineering designs.
Seemingly unrelatedly, progress in fields like large-language models (e.g. ChatGPT) or generative AI (e.g. DALL-E or Google Parti) have made the headlines. But what does that mean for us? Generative AI, on the other hand cannot just produce pretty pictures as show here – it has the potential to transform the way chemical engineers approach the design of new materials, chemicals and even suggest process flowsheets or engineering designs.
Large language models, such as GPT-3, are capable of processing and understanding vast amounts of natural language text, which can be particularly useful where large amounts of technical documentation or patents must be analyzed and understood.
 These models can help chemical engineers to identify trends, predict outcomes, find answers and generate new insights based on a deep understanding of historic data. To underline the potential, this abstract was generated by providing a simple prompt.
These models can help chemical engineers to identify trends, predict outcomes, find answers and generate new insights based on a deep understanding of historic data. To underline the potential, this abstract was generated by providing a simple prompt.
This presentation will give an overview, share examples on these topics and highlight drivers, as well as obstacles and inhibitors of this development to inspire you to explore the field further.
29 June 2023 – Prof. Ivan Kempson, University of South Australia
Prof. Ivan Kempson

Future Industries Institute
University of South Australia
Profile
Mechanistic insights into nanoparticle modulation of therapeutic response to radiation therapy
Metal nanoparticles are in clinical trials and have market approval in Europe. Scope exists to further their mechanistic understanding and improve their action for enhancing the therapeutic ratio in radiotherapy treatment.
We have been investigating nanoparticle structure-function relationships in a radiotherapy context down to the single cell level. To pre-clinically validate two lead nanoparticle formulations in sensitizing triple negative breast cancer to radiotherapy and assess mechanistic action.
A variety of in-vitro studies have been conducted to understand nanoparticle uptake across cell populations (using cross-correlative confocal microscopy and synchrotron X-ray fluorescence), effects on cell metabolism (fluorescence lifetime microscopy), and the nanoparticle-proteome nexus. Radiobiological response studies have advanced to in-vivo studies with immunocompetent Balb/c mice bearing 4T1 tumours as a model of triple negative breast cancer. In a partnership with the Royal Adelaide Hospital we utilise clinical facilities and mice were irradiated with 4x4Gy fractions from a 6 MV linac after systemic administration of nanoparticle formulations functionalised to target cancer cells. Tumour volume and survival were measured for control arms, and radiotherapy treatment with/out receiving nanoparticles. Mechanistic studies monitored metastatic burden in lung and dissociated tumours were characterized at the single cell level with mass cytometry to identify which cell populations the nanoparticles associate and identify manipulation of the tumour cell-composition.
Nanoparticles were associated with a range of cell types despite being targeted to cancer cells and instigated remodelling of the tumour microenvironment. Median time of animal survival improved by 30% and metastatic burden in lungs was reduced by 70% through use of a nanoparticle radiosensitizer in conjunction with radiotherapy compared to radiotherapy alone.
While the initial premise of high atomic number nanoparticle radiosensitization was based on physics theories, the nano-bio interaction appears to ultimately dominate radiobiological response.
Clinical use of nanoparticle radiosensitizers is likely to increase and understanding their mechanistic modes of action will aid in optimizing formulations to promote the best clinical outcomes.
Further Reading: Kempson I. Mechanisms of nanoparticle radiosensitization WIREs Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021, 13, e1656.
20 July 2023 – Prof. Dr. habil. rer. nat. Sabine Enders, KIT (N.B. different time and location!)
Prof. Dr. habil. rer. nat. Sabine Enders

Institut für Technische Thermodynamik und Kältetechnik
KIT
Institute website
Time-dependent interfacial properties
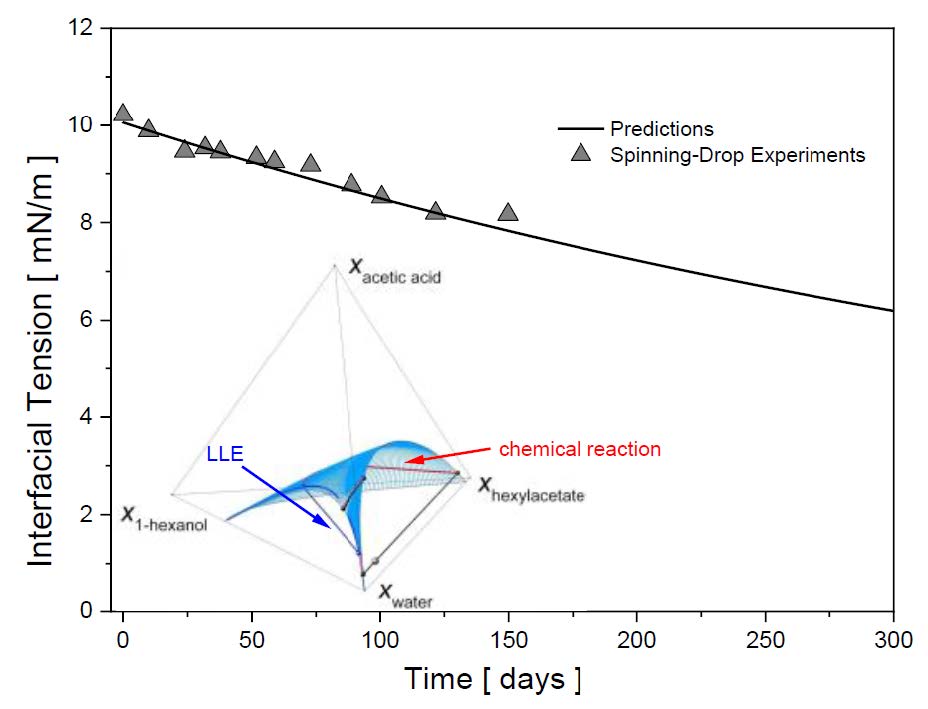
The interfacial properties of mixtures are essential for the development or improvement of separation processes. These properties, at phase equilibrium, can be calculated using the density gradient theory (DGT) in combination with a suitable thermodynamic model. However, in the case of reactive extraction or reactive distillation, the interfacial properties change with time caused by the chemical reaction taking place. This situation requires a new approach that considers the superposition of the chemical and the phase equilibria, as well as kinetic aspects. Recently, a new theoretical framework for the calculation of time-dependent interfacial properties was developed. The new theoretical framework was applied to reactive extraction based on the very slow esterification reaction of 1-hexanol with acetic acid to hexyl acetate and water, and allows the prediction of the interfacial tension as a function of time (Figure 1).[1] The same mixture can also be studied for reactive distillation.
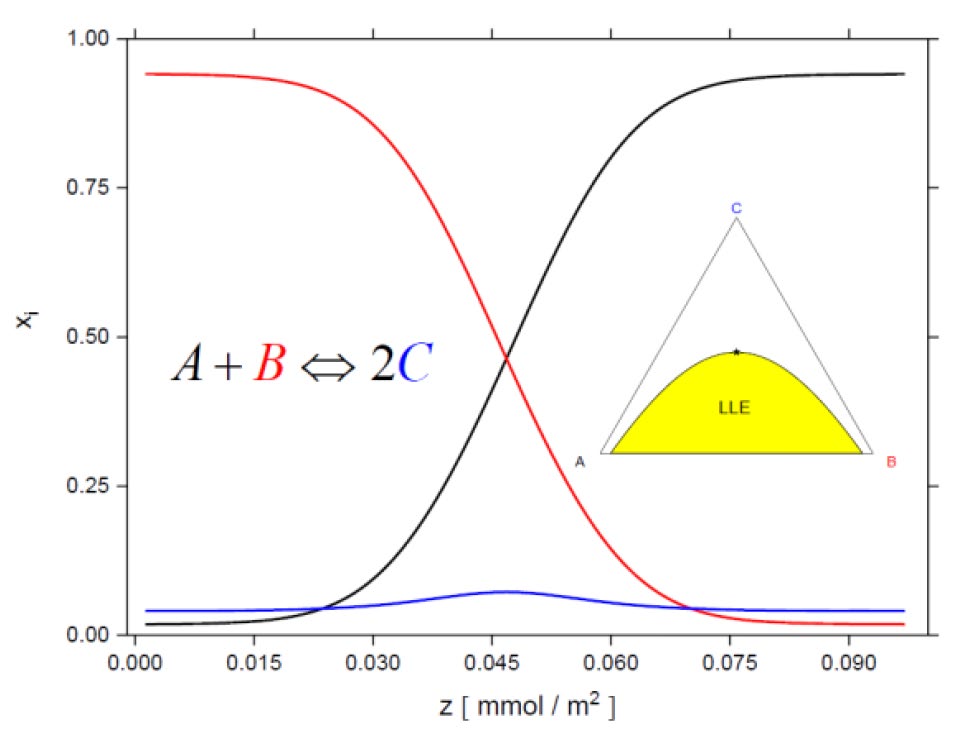
As an extended feature, the DGT allows the calculation of concentration profiles with time- and spatially-resolved resolution within the interface. The predicted concentration profiles, which often contradict the assumption of the classical two-film theory, can be used to optimize the material transport between two phases. For fast chemical reactions taking place in the demixed mixture, the reaction product will be formed only at the interface.[2] One example is depicted in Figure 2.
References
[1] A. Danzer, S. Enders, The Modelling of the Time-Dependency of Interfacial Properties due to Chemical Equilibrium Reactions in Demixed Fluid System. Fluid Phase Equilibrium 499 (2019) 112240.
[2] J. Hajjar, S. Enders, Interfacial properties of fast chemical reactions occurring in demixed ternary mixtures. Fluid Phase Equilibria 568 (2023) 113758.
15 December 2022 – Prof. Dr.-Ing. Alexander Grünberger, KIT

Prof. Dr.-Ing. Alexander Grünberger
KIT
Research Profile
Colloquium video (contact organizer for access)
Microfluidic single-cell cultivation: A key for next generation bioprocess development
Our understanding of large-scale bioprocesses is still dominated by an average cell approach in which the reactor is well-stirred and all cells behave equally. In contrast, the complex interplay between environmental reactor dynamics and the subsequent cellular response is still a mystery. As a result, cells often show a significant variability in growth and production, potentially influencing the performance and robustness of bioprocesses [1]. Therefore, novel analytical methods need to be developed to understand these processes in detail [2,3]. In this presentation I will give an introduction into bioprocess microfluidics especially microfluidic single-cell cultivation and its application for bioprocess engineering, biotechnology and life sciences. Microfluidic single-cell bioreactors [4] offer precisely controlled external environmental conditions and allow single-cell analysis at full spatio-temporal resolution. I will demonstrate the versatility of these reactors and how these can be used to get a deeper understanding of microbial bioprocesses on the example of industrially relevant production hosts [5-8]. Latest developments, results as well as technological challenges will be discussed.
References
[1] Delvigne et al., (2017), Journal of Biotechnology, 12(7):1600549
[2] Grünberger, A., et al., (2014), Current Opinion in Biotechnology, 29:15-23
[3] Dusny, C and Grünberger, A., (2020), Current Opinion in Biotechnology, 2020, 63:26–33
[4] Grünberger, A., et al., (2012), Lab on a Chip, 12(11): 2060-2068
[5] Mustafi et al., (2014), PlosOne, 9:e85731
[6] Täuber et al., (2020), Lab on a Chip, 20(23):4442-4455
[7] Ho et al., (2022), Frontiers in Chemical Engineering, 4:826485
[8] Täuber et al., (2022), Biotechnology and Bioengineering, DOI: 10.1002/bit.28208
19 January 2023 – PD Dr. Andreas Vorholt, Max Planck Institute for Chemical Energy Conversion

PD Dr. Andreas Vorholt
Max Planck Institute for Chemical Energy Conversion
Leader of Multiphase Catalysis Group
Research Profile
Colloquium video (contact organizer for access)
Operando NMR and FTIR for a look below the surface: deactivation mechanisms in continuous operated processes
In this talk the design of stable catalyst systems in continuous operated miniplants will be discussed.
In a first example the reductive hydroformylation of Fischer Tropsch products will be introduced. A catalyst development and campaigns in miniplants setups will be shown to proof the stable catalyst recycling. These alcohols can be applied in EN590 compatible diesel fuels.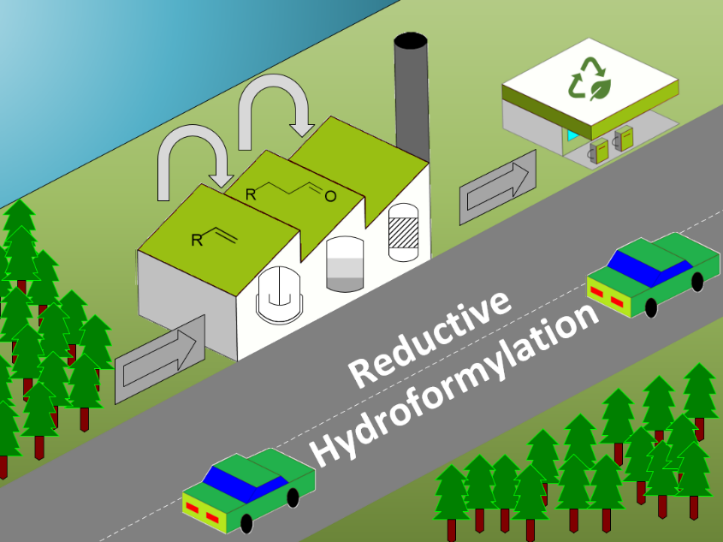
A further topic is to understand the impact of interfacial area in liquid/liquid systems on the catalytic performance. The interfacial area was recorded for the first time during a reaction. This dynamic behaviour was described for the first time and give an insight on the intensification of liquid/liquid techniques.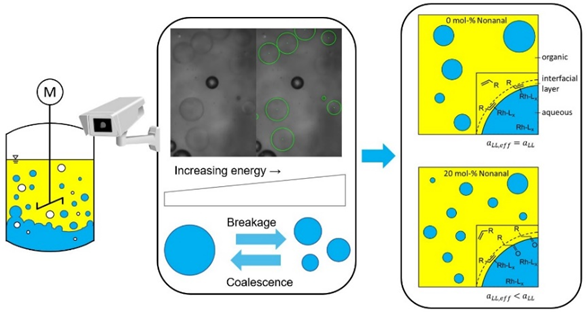
In the last part catalyst deactivation, a frequently observed challenge in molecular catalytic systems, is investigated in a continuous operated miniplant. Transmission FTIR spectroscopy, is a useful tool in detecting catalysts in hydroformylation. Also, the application of Flow NMR has become more widespread in recent years and has already shown its usability in exploring new deactivation pathways in transfer hydrogenation reactions. Using operando transmission FTIR and NMR spectroscopy ligand degradation mechanisms that have previously been only studied with ex situ methods were revised, with ligand oxidation or P-C-bond cleavage as examples.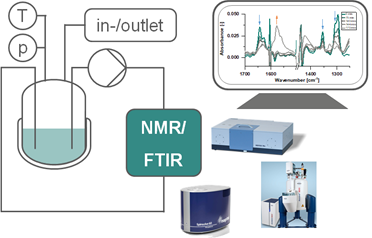
More specifically, the Pd-catalysed oxidation of phosphine ligands is such a deactivation mechanism in molecular catalysis [5] and can be tracked via 31P NMR and FTIR spectroscopy. It can be shown that both NMR and FTIR spectrometry are suitable methods to follow the oxidation of triphenylphosphine to triphenylphosphine oxide a classic deactivation pathways.
26 January 2023 – Prof. Dr. Ulrich Tallarek, Philipps-Universität Marburg

Prof. Dr. Ulrich Tallarek
Philipps-Universität Marburg
Department of Chemistry
Head of Functional Porous Solids group
Research Profile
Colloquium video (contact organizer for access)
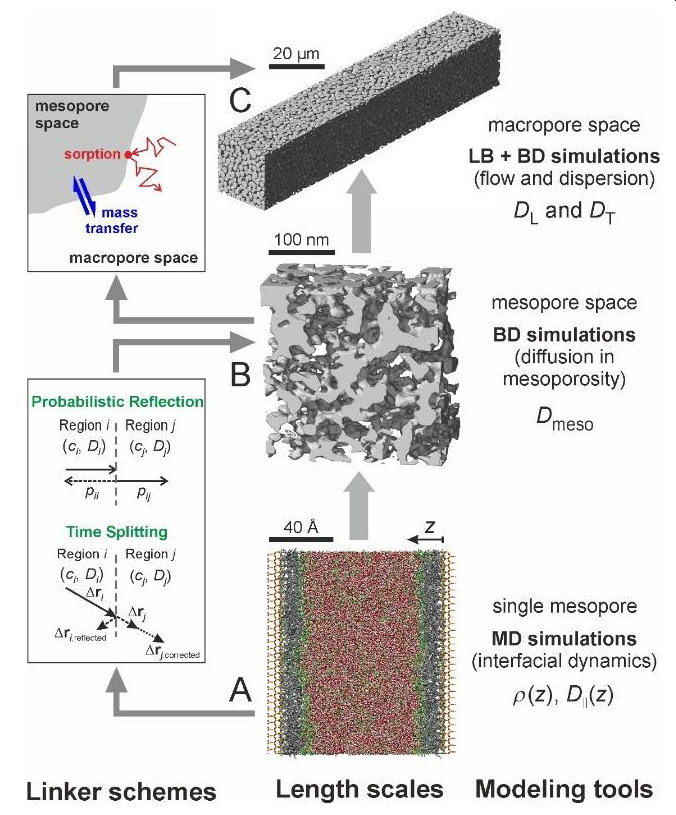
Multiscale simulation of solute sorption, diffusion, and advection in hierarchical macro-mesoporous materials
A bottom-up approach to the simulation of solute transport in hierarchical, macro–mesoporous adsorbents is presented and illustrated for reversed-phase liquid chromatography, the most important chemical separation and purification technique in academic and industrial settings (Figure 1). Gained insight promises a to date unknown understanding of how the interfacial dynamics affects macroscale transport and chromatographic separation efficiency as a function of the fluid flow velocity. In particular, the approach allows to assess individual contributions to longitudinal and transverse dispersion coefficients and related chromatographic plate heights. These include (i) sorption and partitioning at the surface, (ii) effective diffusion in the mesopore space, and (iii) fluid flow and the engendered hydrodynamic dispersion in the macropore space. Efficient linker schemes guarantee the integration of the (detailed molecular-level) interfacial dynamics into the complex hierarchical porosity and multiscale transport models.1,2
Literature:
(1) U. Tallarek et al., J. Phys. Chem. C 2019, 123, 15099–15112.
(2) U. Tallarek et al., J. Phys. Chem. C 2022, 126, 2336–2348.
2 February 2023 – CBI Symposium (starting 4:00 p.m.)
The CBI symposium provides a platform for the scientific exchange between researchers and students. Doctoral candidates will present their current research through posters. Meet the scientists and gain valuable insights into the current developments in the fields of Chemical and Biological Engineering. You are warmly invited!
Agenda
- 4:00 Welcome
- 4:05 CBI at a glance
- 4:15–4:35 Dr. Carlos Bassani (MSS), „Multiscale modeling and simulation of materials and processes“
- 4:35–4:55 Dr. Alexandra Inayat (CRT), „Towards greener syntheses of porous materials“
- 4:55–5:15 Prof. Dr. Katharina Herkendell (EVT), „Bioelectrocatalytic energy systems“
- 5:15–5:35 Prof. Dr. Tanja Franken (CRT), „Catalysts for dynamic CO2 Methanation“
- From 5:30 Poster Session & Awards Ceremony
5 May 2022 – Prof. Jennifer Sinclair Curtis, University of California, Davis

Prof. Jennifer Sinclair Curtis
Distinguished Professor of Chemical Engineering
University of California, Davis
Research Profile
Colloquium video (contact organizer for access)
Toward Simulation-Based Design of Particle Handling Processes
Particulate processes pervade the petroleum, chemical, mining, pharmaceutical, and agricultural industries. Many of these processes have significant opportunities for optimization and productivity enhancements. Reliable particle modeling and simulation tools can improve critical understanding and design of particle transport processes. One approach to modeling particle flow employs the discrete element method (DEM). In DEM, the dynamics of individual powder particles are described rather than treating the particle phase as a continuum via Computational Fluid Dynamics (CFD). DEM models are especially useful for investigating phenomena occurring at the scale of an individual particle. Although DEM models are computationally intensive, they are also capable of developing constitutive relations for the particle phase that are needed for continuum-based CFD simulations. Some examples of application of DEM modeling to complex particulate flows will be discussed, as well as their validation via experiments in hoppers, agitated mixers, and shear cells.
28 October 2021 – Prof. Leopoldo Gómez, Universidad Nacional del Sur
Prof. Leopoldo Gómez
Department of Physics, Universidad Nacional del Sur, Argentina
Humboldt Fellow, Institute of Multiscale Simulation, FAU
Research Profile (ResearchGate)
Colloquium video (contact organizer for access)

Packing Structure of Entangled Filamentous Matter
While linear polymers are well understood, the structure and dynamics of polymer rings remain elusive. Key concepts such as confining tubes and reptation, which are the cornerstone of polymer physics, could be misleading in the ring dynamics. Here we use commercial rubber bands imaged through X-ray tomography to compare the structural properties of semiflexible linear and ring assemblies. Individual band configurations in the assemblies are obtained through a machine learning segmentation
approach, allowing the detailed geometrical and topological characterization of the structures. Assemblies of short rings assume a liquid-like disordered structure, with short-range orientational order, and reveal only minor influence of the container. In the case of longer rings, the confinement causes folded configurations and the bands interpenetrate and entangle. Although rings tend to be more compact and display higher curvature, the topology of rings and linear assemblies are shown to be rather similar under confinement, when scaled properly.
Prof. Leopoldo Gómez
Department of Physics, Universidad Nacional del Sur, Argentina
Humboldt Fellow, Institute of Multiscale Simulation, FAU
Research Profile (ResearchGate)
Colloquium video (contact organizer for access)
4 November 2021 – Prof. Dr. Bastian Etzold, TU Darmstadt (Joint colloquium with CRC 1452)

Prof. Dr. Bastian Etzold
Institute of Chemical Technology
TU Darmstadt
Group Website
Colloquium video (contact organizer for access)
Handshake of Material Development and Electrochemical Process Engineering
in H2 Energy Storage and Power-to-X
The energy supply and chemical industry are a cornerstone of today’s society but both are currently based on fossil resources. Defossilization of both sectors will be vital for the energy transition as well as establishing a circular economy. Sustainable electricity from wind power and photovoltaics will drive these transitions and the technologies of water electrolysis and fuel cells will be indispensable for storage and release of this renewable energy. Furthermore, electrochemical synthesis is highly attractive as it allows direct use of renewable energy to drive chemical production and can employ sustainable feedstock like biomass or CO2. In the future electrochemical processes for Energy and Power-to-X will thus gain tremendously importance.
These new electrochemical processes differ strongly from current heterogeneously catalyzed gas phase bulk chemical production or combustion power plants. Thus, major scientific and engineering challenges need to be solved for technical realization of these novel processes and realizing high energy and feedstock efficiency, as well as space-time-yield. Interdisciplinary research of process engineers, chemical engineers, and technical chemists from the atomic scale towards the full process scale therefore needs to go hand in hand. The lecture will give examples into actual research of the etzoldlab at these different scales. Insights into the ionic liquid modification of electrocatalysts for fuel cells will be given and expanded towards the need of full electrode preparation and testing at industrial reaction rates, which are orders of magnitude higher, compared to classical electrochemical lab testing [1]. The challenges of experiments at industrial operation rates will be discussed and the development of a new experiment bridging between idealized lab and full cell testing will be presented. Testing at these high rates will show that besides catalyst development the reaction-transport-nexus plays a dominant role and optimizing transport pathways in electrode design is of similar importance [2]. Finally, an outlook on future assessment of economics and tolerances to energy and feedstock fluctuations of chemical processes through automatized high throughput process simulation will be given.
References
[1] Emerging Applications of Solid Catalysts with Ionic Liquid Layer Concept in Electrocatalysis, Adv. Funct. Mater. 9 (2021) 2010977
[2] Understanding the Activity Transport Nexus in Water and CO2 Electrolysis: State of the Art, Challenges and Perspectives, Chem. Eng. J. 424 (2021), 130501
11 November 2021 – Prof. Wei Liu, Sichuan University & Engineering Research Center for Alternative Energy Material & Devices (N.B. Talk will be Online only!)

Prof. Wei Liu
Sichuan University & Engineering Research Center for Alternative Energy Material & Devices
Research Profile (ResearchGate)
Advancing Lithium Storage for a Sustainable Energy Future
Global climate change has historically called for transition from consuming fossil fuel to renewable energy sources. Developing efficient and low-cost lithium ion batteries (LIBs) for grid scale and mobile applications (i.e. electric vehicles (EVs), consumer electronics, etc.) have become an essential and pressing issue to ensure sustainable prosperity of human society.
In this talk, the history and working principles of LIBs will be introduced. Special focus will be devoted to the fundamental aspects and recent progressive advancement in the anode side in pursuing higher and higher energy density of LIBs. Silicon-based anode and alkali metal anodes are known to store Li-ions with capacity 10 times higher than prevailing carbonaceous anodes, the main impediments towards application and progresses to date will be introduced and discussed.
Our group studied the intrinsic unstable interface of Si and alkali metals during charge-discharge cell cycling, which are the key impediment for their development. Recent progresses on chemistry and engineering in governing surfaces/interfaces of electrode materials will be presented and discussed: (1) A novel angle to understand dendrite growth during alkali metal plating, and the corresponding strategies on “flattening” the deposition morphology; (2) surface coating combined with electrochemical-spectroscopy characterizations to capture the critical role of organic moieties in affecting cycle of anode materials; (3) A 2D materials based scaffold to protect alkali metal anodes towards dendrite-free cycling.
References
(1) Tutorial review on structure – dendrite growth relations in metal battery anode supports, Chemical Society Review, 2020, 49, 7284-7300.
(2) Pristine or Highly Defective? Understanding the Role of Graphene Structure for Stable Lithium Metal Plating. Advanced Energy Materials, 2019, 9, 1802918.
(3) “Sticky” carbon coating enables high-area-capacity lithium storage of silicon-graphitic carbon hybrid Carbon, 2021, 184, 91-101.
(4) Lithium-activated SnS–graphene alternating nanolayers enable dendrite-free cycling of thin sodium metal anodes in carbonate electrolyte, Energy & Environmental Science, 2021, 14, 382-395
13 January 2022 – Dr. rer. nat. Michael Hirscher, Max Planck Institute for Intelligent Systems (N.B. Talk will be Online only!)

Dr. Michael Hirscher
Group leader – Hydrogen Storage
Max Planck Institute for Intelligent Systems, Stuttgart
Research Profile (Publons)
Nanoporous materials for hydrogen isotope separation
One of the important operations in chemical industry is separation and purification of gaseous products. Especially H2/D2 isotope separation is a difficult task since its size, shape and thermodynamic properties resemble each other. Porous materials offer two different mechanisms for separating hydrogen isotopes, either confinement in small pores, i.e., “kinetic quantum sieving”, or adsorption on strong binding sites, i.e., “chemical affinity quantum sieving”. Recently, several new classes of nanoporous materials have been developed, which for the first time allow the exact tailoring of pore size and aperture as well as including open metal sites into the framework.
Experimentally, the measurement of the selectivity for hydrogen isotope separation is very challenging, since it requires low temperatures near the boiling point of the gases. Using low-temperature thermal desorption spectroscopy (TDS), we have developed a method for measuring directly the isotope selectivity after exposure to H2/D2 mixtures.
Exemplarily, this talk will demonstrate the potential of novel nanoporous materials for hydrogen isotope separation on experimental results for metal-organic frameworks (MOFs) and organic cage molecules.
3 February 2022 **Postponed to a later semester** – Prof. Dr. Olga Vinogradova, Moscow State University (Joint colloquium with CRC 1411)

Prof. Dr. Olga Vinogradova
Laboratory of Microfluidics and Nanofluidics
Laboratory of Physical Chemistry of Modified Surfaces
Moscow State University
Group Website
Manipulation of flows and particles in microfluidic devices using lubricant-infused unidirectional surfaces
Lubricant-infused surfaces have opened a whole new field of investigation, with both fundamental and practical perspectives. In my talk, I will first discuss the enhanced slip properties for a liquid flow over lubricant-infused unidirectional surfaces. This situation reflects many practical settings involving liquid flows past superhydrophobic grooves filled with gas, or past grooves infused with another, immiscible, liquid of smaller or equal viscosity. I will then discuss principles of a generation of secondary transverse shear flow near such slippery lubricant-infused surfaces and its possible applications for mixing and separation of particles in microfluidic devices.
31 October 2019 – Prof. Dr. Mischa Bonn, Max Planck Institute for Polymer Research

Prof. Dr. Mischa Bonn
Department of Molecular Spectroscopy
Max Planck Institute for Polymer Research
Molecular Structure and Dynamics of Water and Ice Interfaces or Why Ice is Slippery
Water and ice surfaces and interfaces are ubiquitous, not just in nature (e.g. for various naturally occurring forms of snow and ice) but also in many technological applications (such as food science and artificial snow machines). Water is a rather unique liquid, owing to its strong intermolecular interactions: strong hydrogen bonds hold water molecules together. At the surface of ice, the water hydrogen-bonded network is abruptly interrupted, conferring distinct properties on the interface compared to bulk. We elucidate the structure of interfacial water molecules at the surface of solid ice, and of water in contact with different materials, using surface-specific vibrational spectroscopy of interfacial water molecules. For ice, we find an excess of hydrogen bonds at the ice-vapor interface around 200 K, due to a competition between entropic and enthalpic contributions to the free energy. Around 250 K temperatures, surface melting of ice is found to occur in a bilayer-by-bilayer fashion. Finally, we relate the temperature-dependent molecular structure of the ice surface to the macroscopic friction coefficient, and explain why ice is most slippery around 264 K.
7 November 2019 – CBI Symposium
Note different time (16:00) and location (Seminar room of the IZNF, Cauerstr. 3)
Program:
- 16:00 Welcome from the head of the department, Prof. Dr.-Ing. Stefan Will
- 16:05 Introduction to CBI from the department administrator, Dr. Marlene Reuschel
- 16:15 Dr. Thomas Koller (AOT-TP), „Interfacial Tensions and Viscosities in Multiphase Systems by Surface Light Scattering and Molecular Dynamics Simulations“
- 16:30 Dr. Dominik Müller (EVT), „Ansatzpunkte für künstliche Intelligenz im Bereich von Thermoprozessanlagen“
- 16:45 L. Anschütz, T. Freiwald, A. Kick, H. Rachwalsky, B. Worgul, „Recycling von Smartphones – ein Beitrag zum chemPLANT Wettbewerb 2019“
- 16:55 Dr. Alberto Leonardi (MSS), „Engineered Nanostructured Materials: Coupling Experiment and Simulation“
- 17:10 Dr. Martin Vielreicher (MBT), „Konstruktion von Knochengewebe: Quantifizierung von Kollagennetzwerken und die Aufklärung relevanter Signalwege und mechanischer Einflußgrößen“
- From 17:30 Poster session with refreshments followed by award of poster prizes
21 November 2019 – Dr. Michael Kleiber, thyssenkrupp Industrial Solutions AG
An Azeotrope in the Desert
In polybutylene terephthalate (PBT) production, tetrahydrofurane, a widely applied solvent in chemical and pharmaceutical industry, occurs as a by-product due to dehydration of 1,4-butanediol. It is an attractive option to recover it from a waste water stream.
The separation of the azeotropic binary system tetrahydrofurane / water is a very elegant application of phase equilibrium thermodynamics. Due to the relatively strong pressure dependence of the azeotropic composition, tetrahydrofurane / water is one of the few systems where a simple pressure swing distillation is possible, without introduction of an additional selective agent into the process. This technique was applied in a project on the Arabian peninsula, supplemented by two additional distillation columns for separating high- and low-boiling impurities.
While the general results were quite as expected, it turned out to be not possible to keep the specification for the tetrahydrofurane, which was obviously contradictory to thermodynamics. The presentation describes the way how to find out where the errors occurred. Not only thermodynamics but also analytics, heat exchanger design, sampling, column hydrodynamics, distillation column control, chemistry, meteorology, and, last but not least, Arabian culture had to be taken into account.
Rigorous application of thermodynamics and the knowledge of its capabilities and limitations were finally decisive to come to a solution and avoid unreasonable modifications of the plant.
28 November 2019 – Prof. dr. ir. Marjolein Dijkstra, Universiteit Utrecht
Birth of a Binary Crystal Nucleus of Hard Spheres
Photonic crystals have numerous applications ranging from optical waveguides to ultra-fast optical computers. One way to fabricate photonic crystals is via colloidal self-assembly. The diamond and pyrochlore lattices are the most sought-after structures because of their wide photonic bandgaps. However, these open lattices are difficult to self-assemble as they are mechanically unstable for simple repulsive colloids. To find away around, one can self-assemble both the diamond and pyrochlore structure in a close-packed MgCu2 Laves phase (LP) from a binary colloidal dispersion. By removing the large/small spheres (Mg/Cu) from the MgCu2 phase, one can readily obtain the diamond/pyrochlore lattice. The three LPs, MgCu2, MgNi2, MgZn2, can be assembled in a binary hard-sphere mixture. As all three LPs pack with the same density, the free-energy difference between them is very small, with MgZn2 the most stable phase. Despite this, LPs have never been seen to crystallize in such a mixture in simulations and experiments on colloids with sizes that open up a photonic bandgap in the visible. Using simulations we show that binary hard-sphere mixtures get arrested into a glass, thereby preventing LP nucleation, due to the presence of fivefold clusters in the supersaturated fluid. Our results reveal that by introducing softness in the interparticle potential, one can postpone the glass transition by reducing the concentration of fivefold structures in the fluid phase in such a way that crystal nucleation can proceed without interruption. This finding enabled us to show the first ever instance of spontaneous LP nucleation in nearly hard spheres. Unfortunately the most stable phase in bulk is the MgZn2 phase, and not the aimed MgCu2. Here, we show by simulations that up to one hundred thousands of these particles in a spherical confinement spontaneously crystallize into a 3D icosahedral binary quasicrystal consisting of twenty tetrahedral domains with MgCu2-like symmetry. Finally, we investigate one of the many open questions on icosahedral quasicrystals — Can we watch the birth of an icosahedral quasicrystal? Our simulations show for the first time the birth of an icosahedral quasicrystal of hard spheres.
23 January 2020 – Prof. Daniele Marchisio, Politecnico di Torino

Prof. Daniele Marchisio
Research group on Multiscale Modeling for Materials Science and Process Engineering
Department of Applied Science and Technology
Politecnico di Torino
Group Website
Application of multiscale modelling and deep learning tools for flash nanoprecipitation and reactive crystallization
Crystallization and precipitation are very important in chemical engineering as numerous particulate products are manufactured by relying on these processes. Examples span from the pharmaceutical industry to the materials industry, including catalysis and batteries. The final products properties are characterized in terms of distributions, notably the size distribution. In turn the particle size distribution (PSD), or crystal size distribution (CSD), is strongly affected by the operating conditions under which the process is carried out and in order to design, scale up and down and optimize the process, computational models are extremely useful. In fact, they can answer the complex question: under what operating conditions a specific PSD or CSD can be obtained? In this contribution the main computational modelling techniques are presented and discussed.
As the main emphasis is on the prediction of the PSD/CSD the most popular computational models are based on the solution of the population balance equations (PBE), that dictates the evolution of the PSD/CSD. However, since the PBE is strongly connected with the fluid dynamics of the crystallizer, very often the PBE is tightly coupled with a computational fluid dynamics (CFD) model. The coupling is often realized with quadrature-based moments methods (QBMM) such as the quadrature method of moments (QMOM) or the conditional quadrature method of moments (CQMOM) [1].
Moreover, as the rates with which particles and crystals are formed are governed by molecular processes, very often these models are coupled also with atomistic and molecular models. Among the different modelling choices full atom molecular dynamics (MD) and coarse-grained molecular dynamics (CGMD) are mostly employed.
A plethora of codes is used to run these simulations, to couple the different models and to orchestrate the workflows, ranging from Ansys Fluent, OpenFOAM, code_saturne, LAMMPS, GROMACS, Salome, etc.
All the above-mentioned models, being based on first principles and physical laws are labelled as physics-based models. However, recently the simulation of these processes also relies on the use of artificial intelligence and deep learning tools to build data-driven models, which is contrast to the previous ones, are not based on first principles. An interesting idea is that since very often the amount of experimental data is often not enough to build a data-driven model, validated physics-based models can be used to augment and enrich a limited experimental data set and to build the data-driven model, following the digital twin concept.
Two examples will be discussed in this presentation. The first one focuses on flash-nanoprecipitation via solvent-displacement for the production of amorphous polymer nanoparticles for controlled drug delivery applications, whereas the second focuses on the precipitation of inorganic crystals via reactive crystallization or precipitation. In the first example it will be shown how multiscale modelling can be used to simulate the polymer particle formation process, starting from molecules up to the actual crystallizer and to successfully predict the effect of the choice of the good solvent, of paramount important in these processes [2,3]. In the second example the use of these computational tools will be discussed for a different application process involving inorganic crystals.
References
[1] Marchisio D.L., Fox R.O. Computational models for polydisperse multiphase and particulate flows (2013) Cambridge University Press, Cambridge, UK.
[2] Lavino, A.D., Banetta, L., Carbone, P., Marchisio, D.L. Extended Charge-On-Particle Optimized Potentials for Liquid Simulation Acetone Model: The Case of Acetone-Water Mixtures (2018) Journal of Physical Chemistry B, 122 (20), pp. 5234-5241.
[3] Lavino, A.D., Di Pasquale, N., Carbone, P., Marchisio, D.L. A novel multiscale model for the simulation of polymer flash nano-precipitation (2017) Chemical Engineering Science, 171, pp. 485-494.
6 February 2020 – Dr. Jürgen Pfeiffer, Wacker Chemie AG

Dr. Jürgen Pfeiffer
Director of Battery Materials Division
Wacker Chemie AG
On the Way to Silicon-based Lithium-Ion Battery Anode Materials
The market for lithium-ion batteries has seen a continuous growth since their introduction by SONY in 1991. An addition boost in the last years results from the enormous demand from the development of electric vehicles. However, the adoption is still low because of the limited energy density, i.e. driving range. Based on it’s extremely high lithium storage capacity compared to nowadays used graphite, silicon is one of the most promising next-generation anode materials for lithium-ion batteries especially for electric vehicles. However, the technical utilization of silicon is still hampered by challenges resulting from the huge volume change accompanied with the (de-)lithiation (up to 300 %). These challenges include a) electrochemical milling of the active material, b) electrode damage by binder failure, and c) continuous loss of mobile lithium by (re-)formation of the SEI. Two major strategies to solve these problems including their potential and limitations will be discussed. The first approach includes the modification of the electrode architecture to stabilize the active material, the electrode and the SEI. This requires improved binders and electrolytes. The second strategy is aiming at the development of a new class of active materials that accommodates the volume change of the silicon not on the electrode but on the material level. This can be achieved by introducing the silicon into a porous matrix avoiding direct contact between silicon and the electrolyte.

2 May 2019 – Prof. Ilja Siepmann, University of Minnesota
Predictive Modeling of Adsorption and Transport in Nanoporous Materials: From High-Throughput Screening to First Principles Simulations
Nanoporous materials, such as zeolites and metal-organic frameworks, play numerous important roles in modern oil and gas refineries and have the potential to advance the production of fuels and chemical feedstocks from renewable resources. The performance of a nanoporous material as separation medium and catalyst depends on its framework structure and the type or location of active sites. To date, more than 200 zeolite framework types have been synthesized and more than 330000 thermodynamically accessible zeolite structures have been predicted, and the corresponding numbers are significantly larger for metal-organic frameworks. Hence, identification of optimal nanoporous materials for a given application from the large pool of candidate structures is attractive for accelerating the pace of materials discovery. Here we identify, through a large-scale, multi-step computational screening process, promising nanoporous materials for (i) purification of ethanol from fermentation broths, (ii) sweetening of highly sour natural gas mixtures, and (iii) hydroisomerization of linear to slightly branched alkanes with 18-30 carbon atoms. This talk will also highlight recent developments enabling first principles Monte Carlo (FPMC) simulations for which the potential energy is calculated on-the-fly using Kohn-Sham density functional theory. Applications of FPMC to the prediction of (a) adsorption isotherms for gas molecules in metal-organic frameworks with under-coordinated metal nodes and (b) reaction equilibria in cation-exchanged zeolites. Emphasis will be given to simulation methodologies and microscopic-level origins of the observed thermodynamic behavior.
9 May 2019 – Dr. Jan Saam, OSPIN GmbH
A Modular Platform for the Scalable Automation of Bioprocesses
In this talk a modular and universally applicable platform for the automation, analysis and documentation of bioprocesses will be presented. This platform includes hardware components from which a wide variety of devices and systems can be built, as well as a sophisticated cloud solution for bioprocess design, documentation and data analysis. This enables the design and automated execution of complex bioprocesses e.g. in the fields of tissue engineering, stem cell technology, or food technology. Thanks to the open, modular system, the platform can be expanded at any time, including by the user, and new technologies can be integrated. The OSPIN platform accelerates development times and makes it straightforward to scale customer projects from the R&D phase to industrial applications. This brings the OSPIN platform to the level of an „operating system“ for biotechnology with a wide variety of potential applications.
16 May 2019 – Prof. Dr. Regina Palkovits, RWTH Aachen
Heterogeneous Catalysis and Electro-Catalysis – Crucial Elements of an Efficient and Flexible Use of Renewable Carbon Resources
Renewable carbon feedstocks such as biomass and CO2 present an important element of future circular economy. Especially biomass as highly functionalized feedstock provides manifold opportunities for the transformation into attractive platform chemicals. However, these resources require novel paradigms in process design. Fossil feedstocks are processed in stationary gas-phase processes at elevated temperature. On the contrary, biorefineries are based on processes in polar solvents at moderate conditions to selectively deoxygenate the polar, often thermally instable and high-boiling molecules. Considering “green electrons” provided by renewable energy technologies, also dynamic (electro)catalytic processes become attractive as key technology of a throughout circular economy.
Herein, novel concepts in catalyst design will be discussed for CO2 activation, novel biomass transformations as well as the future role of a potentially electrified biorefinery.
23 May 2019 – Prof. Dr. Michael Sommer, TU Chemnitz
Defective conjugated polymers for organic electronics
Recent years have witnessed tremendous progress in the synthesis of novel, structural diverse conjugated polymers with high figures of merit for organic solar cells, transistors and other applications. Issues pertaining to these materials made by step growth polycondensation is the role of chemical defects that eventually form during synthesis, batch to batch reproducibility and molecular weight control. I will present several examples of conjugated polymers made in our group in which these aspects are correlated with device performance. Using selected examples of naphthalene diimide-, diketopyrrolopyrrole- and benzothiadiazole-based copolymers as p- and n-type materials, application in organic photovoltaics, n-channel transistors and thermoelectrics will be presented, and the role of the mentioned molecular parameters will be discussed. 
4 July 2019 – Prof. Dr. Oliver Hayden, TU Munich
Biomedical Electronics @ TranslaTUM
TranslaTUM is the flagship project on translational cancer research of TUM and a central institute for close cooperations between engineering and clinical sciences at the campus of Klinikum rechts der Isar. The Chair of Biomedical Electronics located at TranslaTUM is mediating the expertise of engineering faculties and supports the development of a translational research between the disciplines and working cultures. Focus of the Chair is to to develop methods for cell function analysis for both pre- and clinical applications. During my talk I will discuss the challenges and opportunities for an academic freshman in such an environment. Last, I will highlight recent project results with potential translational impact to demonstrate our “engineering” strategy for cooperative research at TranslaTUM.

15 November 2018 – Prof. Dr. Gregor D. Wehinger, TU Clausthal
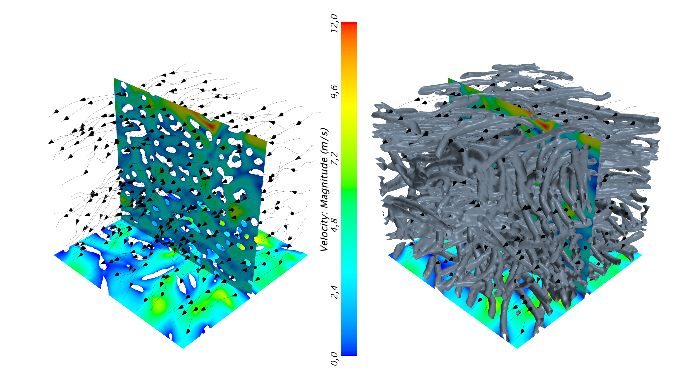
Fixed-bed reactor models with particle-resolved CFD – Pain or gain?
A catalytic fixed-bed reactor is a widely used reactor type in the chemical and process industry. It is a tube filled with catalytically active pellets with gaseous reactants flowing through the bed and being converted into products. There exists models for such a reactor on different levels of detail, starting from simple one-dimensional models. However, the interaction between transport phenomena and the progress of reaction vary throughout the bed. Under extreme conditions, e.g., small tube-to-particle diameter ratios and highly endothermic or exothermic reactions, the assumptions of the simple models collapse. With computational fluid dynamics (CFD) it is possible to resolve transport phenomena, i.e., momentum, heat, and mass transport, as well as surface reactions, with a high resolution. Particle-resolved CFD is the approach to resolve geometrically every pellet inside the bed, see Fig. 1, which leads to a huge amount of information.
Here, an introduction and applications of this approach is given. Afterwards, validation cases are presented and discussed. The synthetically generated bed consisting of different shapes will be compared with experimental data. Heat transfer and heterogeneous catalytic systems are validated with spatially resolved data. The simulation results are in very good agreement with the experiments. However, there are still open questions when surface reactions are included in the model. Still, with this approach it is possible to explore “in silico” different designs, for example different shapes of pellets, which have a strong influence on reactor performance. In a concluding outlook, the transfer of this CFD approach to electrochemical systems is given, see Fig. 2.
22 November 2018 – Prof. Allen Liu, University of Michigan
Push and pull: microfluidics for single cell mechanobiology

The proper responses of cells to mechanical stimuli are important in numerous physiological processes. With the development of microsystem engineering tools, controlled and repeatable application of active mechanical input to single cells is becoming more available. Several microfluidic platforms have been developed for mechanotransduction research over the last decade that many focus applying a single mechanical perturbation and often to a population of cells. Here we develop a multilayer polydimethoxysilane (PDMS)-based microfluidic device with the goal of applying controlled aspiration and compression to single cells. Two independent pneumatically controlled channels above the flow channel serve to facilitate cell loading and compression when they are actuated. As a model system of cell and to demonstrate the salient features of our device, we generated water-oil-water double emulsion droplets and demonstrated trapping, aspiration, and compression of double emulsion droplets. More recently, we have combined this with microcontact printing to confine the size of single cells and investigate the effect of static vs. cyclic compressive stress to single cells. Our unique and versatile microfluidic compression device will provide tremendous opportunities for future single cell mechanotransduction studies. At the end, I will also briefly describe our efforts in engineering artificial cells for biomedical applications.
29 November 2018 – Prof. Patsie Polly, UNSW Sydney
Blending Real and Virtual Laboratories in Practical Lessons to Enhance Learning
Providing large numbers of undergraduate students in scientific and medical disciplines with engaging, authentic wet-laboratory experiences is important, but challenging, especially with increasing student enrolments and developments in state-of-the-art equipment/techniques. Virtual laboratories (vLabs) are a potential means to enable interactive learning experiences and have the scope to: enhance the undergraduate student experience by being hands-on, build analytical skills, and promote higher order thinking such as problem solving. We have developed seven vLabs, the Molecular Expression Virtual Laboratory Suite, which are integrated and link to one another to teach technical and theoretical concepts that underlie gene and protein expression, including RT-PCR, qPCR, Western blotting, EMSA, cell culture, SNPs and siRNA. The Molecular Expression Virtual Laboratory Suite exposes students to these experimental techniques, associated analytical skills and the opportunity to learn difficult concepts. Indeed, implementation of the Western blotting vLab (see Figure 1) in a third year Pathology course for science undergraduates has demonstrated its effectiveness in engaging learners. As vLabs are produced on the Adaptive eLearning Platform (AeLP) developed by Smart Sparrow™, students receive immediate formative feedback and staff can track students’ progress and provide remedial action as required. The BEST Network will also be presented to showcase several case studies of how facilitated learning of technical molecular laboratory skills and more routine scientific practical skills can link to development of diagnostic/analytical skills for undergraduate science and medical students. Such skills are important for undergraduates in building a conceptual understanding of translation of laboratory techniques to changes in human biology due to disease. The design and delivery of virtual labs and lessons addresses the issue of evolving learning and teaching paradigms using virtual spaces and also addresses the UNSW 2025 strategic priorities of Academic Excellence (A) and Educational Excellence (A2) particularly across the four pillars that support the Scientia Educational Experience: Communities, Inspired Learning through Inspired Teaching, Being Digital, Feedback and Dialogue.

Short Biography: Patsie is a UNSW Scientia Education Fellow and Associate Professor in Pathology, UNSW Medicine. Recognised nationally and internationally as a medical research scientist, leading higher education teacher and innovative education researcher, she has infused her extensive medical research experience into the classroom by strategically integrating virtual laboratory adaptive lessons, ePortfolio pedagogy and collaborative communities of practice to allow her students to learn career-relevant skills. As UNSW Teaching Fellow, Patsie led course and program-wide ePortfolio implementation/use at UNSW Sydney to facilitate student awareness of professional skills development. Patsie has attracted national funding to support development of eLearning resources. Patsie has been recognised for her innovations in research communication, virtual laboratory lessons and ePortfolio use with numerous institutional/national teaching awards and nominations.
17 January 2019 – Prof. Dr. Felix H. Schacher, Friedrich-Schiller-Universität Jena
Charge control in polymeric materials – from reversible adsorption to catalytically active block copolymer membranes
Well-defined copolymers and block copolymers featuring charge-tunable groups in the side chain are ideal materials for the design and modification of interfaces in various settings[1] or for controlling and directing self-assembly processes.[2] We herein report on the synthesis of polyelectrolytes, polyampholytes, and polyzwitterions as well as the corresponding block copolymers using free radical and controlled radical polymerization techniques. One key building block in our setting is polydehydroalanine (PDha),[3, 4] a polyzwitterion with high charge density and, depending on the pH, tunable net charge (Figure 1).
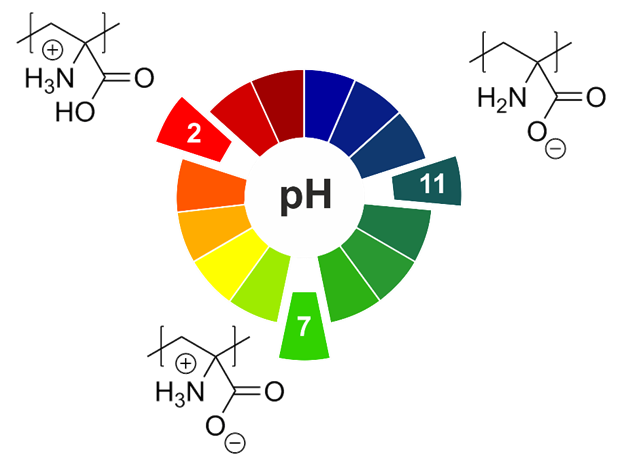
Apart from interesting solution characteristics,[5] we have used PDha and partially protected derivatives as coating materials for iron oxide nanoparticles or within polyelectrolyte multilayers and could show that this allows reversible adsorption / desorption experiments using various oppositely counterparts, including proteins and model pollutants such as methylene blue.[6,7] Besides PDha, we are also interested in the incorporation of polymerizable naphthol derivatives to create polymeric photoacids which show different charge characteristics and solubility in aqueous environment with and without irradiation.[8] Finally, once charge-tunable building blocks are introduced as responsive segment in block copolymer membranes, electrostatic adsorption can be used to immobilize powerful oxidation catalysts such as polyoxometalates based on Mn and V. We could show that such self-supporting hybrid and heterogeneous oxidation catalysts can be used in different model reactions both under static conditions as well as under flow (Figure 2).[9]
References:
1. C. Barner-Kowollik, A. S. Goldmann, F. H. Schacher, Macromolecules 2016, 49, 5001-5016 (Perspective Article).
2. F. H. Schacher, J. C. Brendel, Chem. Asian J. 2018, 13, 230-239.
3. U. Günther, L. V. Sigolaeva, D. V. Pergushov, F. H. Schacher, Macromol. Chem. Phys. 2013, 214, 2202-2212.
4. M. Billing, F. H. Schacher, Macromolecules 2016, 49, 3696-3705.
5. M. Billing, G. Festag, P. Bellstedt, F. H. Schacher, Polym. Chem. 2017, 8, 936-945.
6. M. v. d. Lühe, A. Weidner, S. Dutz, F. H. Schacher, ACS Appl. Nano Mater. 2018, 1, 232-244.
7. P. Biehl, M. V. D. Lühe, F. H. Schacher, Macromol. Rapid Commun. 2018, 39, 1800017
8. F. Wendler, K. Schneider, B. Dietzek, F. H. Schacher, Polym. Chem. 2017, 8, 2959-2971.
9. I. Romanenko, M. Lechner, F. Wendler, C. Hörenz, C. Streb, F. H. Schacher, J. Mater. Chem. A 2017, 5, 15789-15796.
24 January 2019 – Dr. Andreas Schröder, German Aerospace Center (DLR)
Dense 3D Lagrangian Particle Tracking using Shake-The-Box – A new key for turbulence research
The novel 3D Lagrangian Particle Tracking method “Shake-The-Box” (STB) is able to extract particle trajectories from volumetric flow measurements at unprecedented numbers. STB overcomes the ill-posed reconstruction problem for 3D particle distributions at high seeding densities present for each single time-step (as known for tomo-PIV and PTV) by pre-solving the problem for each predicted time-step in a cascade of reduced complexity along a time series of images. It delivers accurate data on particles position, velocity and acceleration (material derivative) along densely distributed tracks. Subsequently, data assimilation approaches can be applied to the dense and scattered input data. An example is the “FlowFit” (FF) method, which uses Navier-Stokes-constraints for an iterative non-linear optimization process in order to gain a continuous interpolation of the flow field with minimized deviation to the measurement data. As a result one gets the full time-resolved velocity gradient tensor and pressure field with high spatial and temporal resolution.
The STB method can be applied as well to short recording sequences, acquired with multi-pulse techniques, allowing investigating high-speed flows at Reynolds numbers relevant for research in aerospace engineering.
Recent developments of STB and FF will be presented here, along with data from accuracy studies using synthetic experiments and a range of experimental applications to turbulent flows in small and large scale volumes (up to cubic-meter-scales using HFSB seeding). The combined availability of a large number of samples of dense 3D Lagrangian particle tracks and the full velocity gradient tensor allows for a multitude of evaluation options, e.g.:
- Extracting instantaneous 3D pressure fields and determining related loads on surfaces
- Investigating the spatial and temporal development of coherent flow structures
- Computing profiles and fields (mean velocity, Reynolds stresses, acceleration) in sub-pixel resolution
- Computing other one- and two-point–statistics (e.g. Joint PDFs, two-point-correlations, dissipation rate) in an adaptive (and conditional) bin-averaging approach
- Determining conditional analysis and statistics of flow events in a combined Lagrangian and Eulerian frame
Examples of the listed approaches for flow analyses and turbulence characterization based on experimental data will be demonstrated.

31 January 2019 – Prof. Dr. Leo van Wüllen, Universität Augsburg
Modern Solid State NMR strategies in Materials Science (or NMR is wunderbar)
For a controlled fine-tuning of materials’ key properties, a detailed knowledge of the structural and dynamic features of the materials poses a prerequisite. To this end we develop and utilize modern Solid State NMR strategies which not only provide information about the structural motifs on short and intermediate length scales but also offer a handle to study the microscopic dynamics within these materials. Especially for materials, which are characterized by a lack of translatorial periodicity, considerable disorder or high dynamics, these approaches offer unprecedented insight into the structure and dynamics. We will show how these novel approaches may be successfully employed to study the structure of amorphous materials at ambient conditions and to obtain information about its evolution with temperature. Further, new solid state electrolytes for next generation’s Li batteries will be presented with the focus here being on the local Li coordination and the mechanism of ion transport. Along the way, a variety of novel NMR (hardware) solutions will be presented, even widening the range of possible applications of NMR spectroscopy in materials science. Examples include MAS NMR at high spinning speeds and ultra high temperature and approaches for high resolution dipolar based NMR spectroscopy without fast MAS.
7 February 2019 – Prof. Dr. Joachim Groß, Universität Stuttgart
Predicting interfacial properties using approaches from molecular thermodynamics
Classical density functional theory (DFT) and molecular simulations are powerful methods for predicting adsorption phenomena and for predicting properties of liquid-fluid interfaces. The talk assesses DFT through comparison to results from molecular simulations and compares both to experimental data. Systems thereby investigated include vapor-liquid and liquid-liquid interfaces of mixtures, as well as adsorption processes at planar walls or microporous materials. Predictions from DFT are found in very good agreement to results from molecular simulations. Contact angles of liquid droplents on solid substrates are also described in good agreement to experimental data. Dynamic density functional theory (DDFT) allows predicting coalescence events of droplets and therewith forms an important bridge to continuum mechanics.
Right: density profile of fluid in a rough micropore
28 February 2019 – Prof. Dr. Ferdi Schüth, Max-Planck-Institut für Kohlenforschung
Beyond oil and gas – Mobility of the future
Oil is a finite resource, and combustion of fossil fuels contributes to global warming. On the other hand, oil is currently the backbone of our mobility system. With increasing concerns about global warming and dwindling resources, we have to develop alternatives on a very large scale. These could be different synthetic liquid fuels, which are accessible from renewable carbon sources, or gaseous energy carriers, such as hydrogen or renewable methane. Moreover, a substantial part of our mobility system could be transformed to electricity for propulsion. In the presentation, options for future mobility systems, obstacles and possible transformation pathways will be discussed.
26 April 2018 – Prof. Magnus Ingelman-Sundberg, Karolinska Institutet
Liver organoids to study liver function, liver disease and drug response
25 % of the global population suffer from nonalcoholic liver disease (NAFLD) and this condition increases very much in recent years. NASH and liver cancer are to a great extent the cause of prolonged NAFLD. In order to obtain new treatment regimens it is important to have relevant human iin vitro systems as the basis for evaluation of mechanisms and treatment options. Hepatic in vitro systems should be able to provide a cellular phenotype similar to the situation in vivo in man. Using novel a model of 3D PHH spheroids we observed that drug metabolism was preserved for several weeks of cultivation and that transcriptomic, proteomic and metabolomics analyses revealed similar phenotype as in freshly isolated hepatocytes. In addition using this 3D spheroid systems we have been able to mimic different liver disease like NAFLD, NASH and fibrosis and found the system suitable for evaluation of mechanisms behind and for identification of drug candidates. In the lecture recent results describing the properties and usefulness of the system will be presented.
3 May 2018 – Prof Sara Skrabalak, Indiana University
Multimetallic Nanomaterials by Design
The importance of molecular structure to molecular function is a central tenet in modern chemistry, with the lock-and-key model of enzyme activation representing a classic example. Likewise, the function of inorganic nanomaterials depends on a number of structural parameters that include crystallite size and shape as well as architecture (e.g., hollow versus solid). To realize the function of such materials, these structural parameters must be precisely controlled and the Skrabalak group is expanding the synthetic toolkit to achieve such advanced nanostructures. This seminar will highlight the use of seed-mediated co-reduction as a route to structurally defined bi- and trimetallic nanomaterials, hierarchical materials, and intermetallic (i.e., ordered alloys) compositions. These synthetic advances, in turn, are enabling previously unimagined nanostructures to be accessed with new function for applications in chemical sensing and catalysis. Ultimately, understanding the relationship between nanostructure form and function will allow this relationship to be inverted to achieve materials by design. Still, the synthetic toolkit must exist to realize this vision and achieve desired nanomaterials on demand.
17 May 2018 – Prof Hans Hasse, University of Kaiserslautern
OME: Process engineering for new synthetic fuels
Developing and realizing sustainable concepts for the future of mobility is presently one of the most urgent tasks. There will be no simple one-cap-fits-all solution for the challenges in this field. Different options will have to be explored and combined suitably. Synthetic fuels play an important role in this. In Germany, much of the current discussion in this field is focused on the NOx and soot emissions of diesel engines. Synthetic fuels can contribute to solving these problems. The most attractive synthetic fuel to replace fossil diesel is OME. Germany is presently the leader in the technology to produce OME. However, it is likely to lose that position to China, where, in contrast to Germany, large-scale plants for producing OME have recently become operative.
OME (oxymethylene dimethyl ethers) are oxygenates of the structure H3C-O-(CH2O)n-CH3. OME fuels strongly reduce the soot formation in diesel engines. As a consequence, also the NOx emissions can be strongly reduced [1,2]. All routes for the production of OME are based on synthesis gas and proceed via methanol. Hence, the raw material basis for producing OME is extremely flexible: either traditional (coal, gas) or renewable raw materials (biomass, CO2) can be used. While older routes for the production of OME involved expensive intermediates like methylal and trioxane [2], this work presents a novel OME process that uses aqueous solutions of formaldehyde and methanol as feedstock. The process design is extremely challen¬ging as OME of the desired chain lengths n = 3 − 5 have to be separated from reactive multicomponent mixtures which show several (reactive) azeotropes, liquid demixing and solid formation. The successful design requires combining advanced experimental techniques like NMR spectroscopy with advanced physico-chemical modeling and process simulation.
The presentation covers all sides of the topic: from the physico-chemical fundamentals, over the experimental studies and the modeling, simulation, and optimization, to the application of OME as synthetic fuel and its perspectives.
7 June 2018 – Prof Stefan Luding, University of Twente
From small-scale particle system simulations to macro-scale theory and applications in civil, mechanical and food processes
The dynamic behaviour of granular materials and powders is of considerable interest in a wide range of industries like geotechnics, food, civil, or chemical/mechanical engineering. However, the full understanding or control of the different phenomena and mechanisms of the particle systems, natural phenomena, or processes is an essential challenge for both science and application due to the multi-scale nature from particle contacts via particles, force-chains, and collective macroscopic flow and structures.
The fundamentals can be studied by direct particle simulation methods, where often the fluid between the particles is important too, in order to gain a microscopic understanding of the processes and mechanisms. For large-scale applications, a micro-macro transition towards continuum theory is necessary, however, only smaller applications can be modeled nowadays directly by discrete micro-scale methods. Instead, more often meso-scale methods are used where the particles are up-scaled, representing a certain number of primary particles. As one example for such meso-models, we use experiments and discrete particle simulations (DEM) to investigate the dosing of cohesive fine food-powders. Other applications involve avalanches or chute flows, ring-shear rheology testing of granular flows as well as the study of the elastic, or elasto-plastic solid-like material behavior.
As alternative multi-scale approach, the micro-macro transition from discrete particulate systems to continuum theory involves a mathematical homogenization or coarse-graining that translates particle-positions, -velocities and -accelerations into density-, stress-, and strain-fields, by statistical spatial- and temporal averaging. The macroscopic fields are compatible with the conservation equations for mass and momentum of continuum theory, and also the fluctuating kinetic energy provides a measure for the importance of fluctuations in those systems. The ultimate goal is to find constitutive relations that contain information about the micro-structure and -fluctuations, and to solve those on the macro-level for solving application and optimization problems. Examples considered are chute-flows down slopes, ring-shear testers as well as rotating drums, where the same local rheology models all should work, independent of the geometry.
14 June 2018 – Prof Marc-Olivier Coppens, University College London
A Nature-Inspired Approach to Process Intensification — Avenues to Scalability, Efficiency and Robustness
Process intensification is key to living organisms. Within the constraints posed by the environment, various properties are needed: scalability, efficient transport across length scales, the emergence of robust and resilient operation by collective processes (in a cell, amongst cells, amongst organisms and their environment), and more. When resources are scarce, efficiency and sustainability are essential for survival. Clearly, all these features apply equally to industrial catalytic processes. However, current processes and materials are often designed using bottom-up principles, matching constraints a posteriori, while applying rational design principles at individual length scales only.
Discovering and applying the principles behind desirable traits in natural systems, like scalability and efficiency, can guide the intensification of chemical processes and the design of novel catalytic and separation systems. We demonstrate the potential of this “nature-inspired chemical engineering” (NICE) approach, in which (1) fundamental mechanisms omnipresent in nature (hierarchical transport networks, force balancing, dynamic self-organization) are utilized instead of imitating a single feature and (2) these mechanisms are properly translated within the context of the technological application.
I will give concrete examples of how the NICE methodology is applied to the intensification of multiphase reactor operation (fluidized beds, in particular) and catalytic processes (including heterogeneous, bio- and electro-catalytic processes, such as fuel cells). Time permitting, I will also touch upon membrane separations.
21 June 2018 – Prof Brij Moudgil, University of Florida
Antimicrobial Photocatalytic Coatings: Pathways from Concept to Market
Titania (anatase polymorph) has been extensively studied as a photocatalyst for organic pollutant degradation as well as microbial inactivation. Commercially, titania has been applied only as self-cleaning coatings mostly in Japan, South Korea and Singapore. Changes in the color and texture of substrates as well as the need for ultraviolet excitation for photocatalytic activity are major limitations for commercial use of titania for antimicrobial and indoor applications. Achieving visible light activation and coating transparency can lead to broader applications of titania as a photocatalyst. We have attempted to achieve these goals by creating a Titania–Polyhydroxy fullerene (PHF) composite coating formulation. In this presentation, processing required for achieving the desired technical specifications, and marketing pathways necessary for commercializing laboratory innovations by a small company will be discussed.
28 June 2018 – Prof Raimund Horn, Hamburg University of Technology
Catalytic Fixed Bed Reactors – Experimental and Numerical Reactor Diagnostics and Knowledge Based Optimization
Catalysis and Reaction Engineering are key technologies for our modern society. Catalytic reactors produce transportation fuels, base chemicals for polymers, fertilizers, and pharmaceuticals, they are used for energy conversion and they clean emissions from mobile and stationary sources. Academic and industrial researchers around the globe optimize existing and search for new catalytic processes to meet current and future challenges. This business is competitive and sometimes frustrating. Promising catalysts often do not perform well outside the lab and improvements to established catalytic processes are incremental. One reason for this is that catalysts are dynamic. Like a chameleon changes color and pattern to adapt to its habitat, catalysts change their structure and reactivity as function of local temperature and concentration conditions in the reactor. While a chameleon still behaves like a chameleon, a catalyst in a technical reactor might behave totally different than in a well defined laboratory testing unit. In most applications, the processes inside the catalytic reactor remain hidden. Reactors are normally non-transparent, operate at high temperature and pressure conditions and contain toxic, flammable or even explosive chemicals. Figuratively speaking, the reactor is a „black box“ (Fig. 1). Measurements are restricted to inlet and outlet flows, mathematical models are often too simplified and reactor optimization is based on trial and error.
The Institute of Chemical Reaction Engineering at Hamburg University of Technology develops experimental and simulation methods to resolve the physical and chemical processes in catalytic fixed bed reactors. The experimental methods range from measurement of concentration and temperature profiles in reactors to spatially resolved operando characterization of the catalyst in the reactor by Raman spectroscopy. In the field of reactor simulation we generate catalyst packings by DEM simulations and compute the flow-, concentration- and temperature field inside the reactor by particle resolved CFD methods (Fig. 2). The knowledge gained from measurements and simulations leads to strategies to improve the performance of the reactor, e.g. by increasing the yield of the target product and minimizing the formation of waste products.
5 July 2018 – Prof Ellen Backus, Max Planck Institute for Polymer Research
A molecular view of water at the SiO2 and TiO2 interface disturbed by flow and photons
19 October 2017 – Dr. Marcelo Carmo, Forschungszentrum Jülich GmbH
Current challenges to develop the next generation of water electrolyzers
Hydrogen is often considered the best means by which to store energy coming from renewable and intermittent power sources. With the growing capacity of localized renewable energy sources surpassing the gigawatt range, a storage system of equal magnitude is required, such as the production of electrolytic hydrogen by water electrolysis. Despite of more than 100 years of experience in alkaline electrolysis systems, and thousands of plants installed all over the world, only a few systems or industries remain, providing the state-of-the-art of this technology today. This is due to the fact that the cost of electrical energy has always remained as an uncomfortable barrier, with electrolytic hydrogen costs not being able to compete with the costs for the production of hydrogen by conventional steam reforming of fossil fuels. Nevertheless, today, increased interest can be observed for PEM water electrolysis technology, and over the past 20 years, new companies and projects have appeared, with new leaders being consequently established in this growing niche. The reason is that PEM electrolysis provides a sustainable solution for the production of hydrogen, and is well suited to couple with energy sources such as wind and solar. The advantages of PEM electrolysis over alkaline electrolysis together with novel R&D approaches can potentially reduce the investment costs of PEM electrolysers. We expect that in the following years, frontier advancements on PEM electrolysis systems will appear, demonstrating a true capacity to ultimately establish hydrogen as a key player in the energy market, and contribute to a future hydrogen economy.
16 November 2017 – Apl. Prof. Dr. Thomas Sottmann, Universität Stuttgart
Self-Assembled Nano-Structured Fluids: Fundamentals and Applications
This lecture fundamentally bases on the eminent tendency of amphiphilic molecules to spontaneously self-assemble. Striving for the thermodynamically stable state the amphiphilic molecules form nanostructures without any input of energy or formulation procedure. Thus – dependent on the type and number of solvents – nanostructured fluids may form ranging from micelles, bilayers, vesicles, lyotropic liquid crystalline phases to microemulsions. The unique properties of microemulsions, namely, the ultra-low interfacial tension, the large interfacial area and the ability to solubilize otherwise immiscible liquids enable numerous applications. Microemulsions can be successfully used as smart reaction media in organic reactions with reagent incompatibility [1] and for the environmentally friendly degreasing of animal skins [2]. Furthermore, the fascinating nanostructure of both bicontinuous and droplet microemulsions has been copied nearly one-to-one to polymeric materials [3]. A promising idea for the preparation of low-cost, polymeric nanofoams is the “Principle of Supercritical Microemulsions Expansion” (POSME) [4]. Within the framework of a BMBF project we were able to precisely track the pressure-induced transformations in brine/CO2/fluorinated surfactant microemulsions combining a stroboscopic high-pressure cell that provides adjustable pressure cycles (1 < p(bar) < 300 and f < 10 Hz) with time-resolved small angle neutron scattering [5]. In another application of microemulsions, a soot-free and NOX-reduced combustion [6] could be obtained burning water-diesel microemulsions in diesel engines. However, the microemulsion has to be formulated on-board just before the injection unit, if one wants to adjust the water content in dependence of the rotation speed. In order to ensure the fast formation of water-diesel microemulsions, their formation kinetics has been studied by TR-SANS combined with a fast stopped-flow set-up [7].
Literature
[1] T. Wielpütz, T. Sottmann, R. Strey, F. Schmidt, A. Berkessel, Chem.-Eur. J. 12, 7565 (2006).
[2] G.R. Pabst, P. Lamalle, R. Boehn, G. Oetter. R. Erhardt, R. Strey, T. Sottmann, S. Engelskirchen, J. Am. Leather. Chem. As. 99,151 (2004).
[3] R. Schwering, L. Belkoura, R. Strey, T. Sottmann, X. Gong, H. T. Davis, SOFW Journal 135, 43 (2009).
[4] R. Strey, T. Sottmann, M. Schwan, DE102 60 815 B4 (2008). M. Schwan, L. G. A. Kramer, T. Sottmann and R. Strey, Phys. Chem. Chem. Phys, 12, 6247 (2010).
[5] R. Strey, A. Nawrath, T. Sottmann, European Patent application WO 2005012466 A1 (2005).
[6] A. Müller, Y. Pütz, R. Oberhoffer, N. Becker, R. Strey, A. Wiedenmann and T. Sottmann, Phys. Chem. Chem. Phys. 16, 18092 (2014).
[7] H.F.M. Klemmer, C. Harbauer, R. Strey, I. Grillo and T. Sottmann, Langmuir 32, 6360 (2016).
23 November 2017 – Dr. Andrea Knauer, Technische Universität Ilmenau
Application of the Microfluid Segment Technology for the Preparation of Noble Metal Nanoparticles
Noble metal nanoparticles (NP) are of great scientific interest due to their unique optical and electronic properties. Since the physical properties of these particle systems strongly depend on the size- and shape distribution within the particle ensemble, most of the potential applications, as e.g. sensing or labeling purposes, demand high sample homogeneities and high yields of the desired particle shape.
One of the most common nanoparticle preparation methods is the wet-chemical bottom-up approach. Here, the syntheses are based on fast occurring redox reactions, in which a metal precursor (as e.g. silver nitrate or tetrachloroauric(III) acid) is reduced by an appropriate reducing agent (as e.g. sodium borohydride or ascorbic acid). It was found that the reduction of the metal species is completed within a time interval of a few milliseconds [1]. Such fast occurring reactions demand for a powerful mixing method, since the mixing rates of conventional methods (like stirring) are in a time scale of a few hundred milliseconds. In this case, concentration gradients can be expected, which will negatively influence the final size distribution of the particle ensemble. Therefore, the nanoparticle syntheses were carried out using a microfluidic system, which is based on a two-phase liquid-liquid plug flow [2]. In a microchannel (d(i) = 500µm), the aqueous reactant solutions are dosed into a carrier stream of perfluorinated alkanes, which results in regular micro fluid segments. Stable convection pattern emerge inside the aqueous segments, whereby the velocities in the flow field can be increased by an increase in the total flow rate. Due to this effective segment-internal mixing, Au/Ag/Au-core/multishell-NP [3], triangular Ag nanoprisms [4], Au nanocubes [5], and Au nanorods with each precisely tunable sizes and optical properties could be obtained. The presented work will focus on the facile adjustability of the physical properties of noble metal NP systems using a droplet based synthesis method.
[1] Polte, J., Erler, R., Thunemann, A. F., Sokolov, S., Ahner, T. T., Rademann, K., Emmerling, F. and Kraehnert, R. (2010). Nucleation and Growth of Gold Nanoparticles Studied via in situ Small Angle X-ray Scattering at Millisecond Time Resolution. ACS Nano, 4, 1076-1082.
[2] Kohler, J. M., Li, S. N. and Knauer, A. (2013). Why is Micro Segmented Flow Particularly Promising for the Synthesis of Nanomaterials? Chem Eng Technol, 36, 887-899.
[3] Knauer, A., Thete, A., Li, S., Romanus, H., Csaki, A., Fritzsche, W. and Kohler, J. M. (2011). Au/Ag/Au double shell nanoparticles with narrow size distribution obtained by continuous micro segmented flow synthesis. Chem Eng J, 166, 1164-1169.
[4] Knauer, A. and Kohler, J. M. (2013). Screening of Multiparameter Spaces for Silver Nanoprism Synthesis by Microsegmented Flow Technique. Chem-Ing-Tech, 85, 467-475.
[5] Thiele, M., Soh, J. Z. E., Knauer, A., Malsch, D., Stranik, O., Muller, R., Csaki, A., Henkel, T., Kohler, J. M. and Fritzsche, W. (2016). Gold nanocubes – Direct comparison of synthesis approaches reveals the need for a microfluidic synthesis setup for a high reproducibility. Chem Eng J, 288, 432-440.
7 December 2017 – Prof. Dr. habil Andreas Dreizler, Technische Universität Darmstadt
Combustion in the Focus of Laser Light
In the context of the transformation of our energy systems the role of future combustion technologies is briefly discussed. To compensate for load variations in highly fluctuating power from wind and solar, improved combustion processes will contribute substantially to the mix of complementary energy conversion technologies. A major task in combustion science and engineering is the development of load and fuel flexible, highly efficient and clean gas turbines and reciprocating engines. This development is increasingly based on a detailed understanding of the underlying mutually coupled physical and chemical processes. In this talk it will be shown how advanced laser diagnostics in combustion systems contributes to a much improved understanding of the interaction between chemical reactions and turbulent flows that are confined by combustor walls.
11 January 2017 – Prof. Dr. Ulrich Jonas, Universität Siegen
Responsive Polymer Architectures – From Hydrogels to Sensors
The term „reconfigurable“ or „programmable“ soft matter represents an emerging concept in science and technology, where the chemical, physical and functional properties of materials can be switched between well defined states by external stimulation, for example by temperature changes or by irradiation with light. Manifold examples are found in the animate nature, such as phototaxis of plants (like the head of the sunflower following the sun), and at present the challenge in research aims at harnessing such powerful behavior in artificial matter by merging the specific responsive functionality of molecules with a complex structural hierarchy.
Towards this aim we have synthesized a range of thermoresponsive, watersoluble polymers with either a lower critical- (LCST) or an upper critical solution temperature (UCST) by controlled radical polymerization methods. By means of the controlled polymerization method (RAFT and ATRP) specific end group functionalizations can be achieved. Upon variation of the temperature these polymers show a characteristic transition of their solubility and consequently of the coil dimensions in the aqueous phase, becoming soluble above their UCST, or insoluble above the LCST due to polymer coil collapse. These polymers show already a complex aggregation behavior in the solution state, which specifically depended on the end group functionalization. Concomitantly, the aggregation state and superstructure formation (micellar and vesicular aggregates) depends on the external thermal stimulation. The different polymers were also endowed with photoreactive groups that allow crosslinking upon irradiation with light. This photocrosslinking strategy enables immobilization of the transient state when changing external conditions.
From these polymers various hydrogel layer architectures could be realized that find application as responsive sensor matrix with high binding capacity in optical sensor platforms based on surface plasmon resonance or Bloch surface waves.
Details of the various synthesis strategies and characterization results will be presented, and a hypothetical model of the structure-behavior relationship will be discussed based on the hydrophilic-hydrophobic balance of the end groups with the polymer chain.
18 January 2018 – Prof. Dr. Oliver Trapp, Ludwig-Maximilians-Universität München
Self-amplifying Reactions: New Perspectives for Origins of Life
Self-amplifying enantioselective reactions are important in the context of Origins-of-Life to understand the formation of a homochiral world. The understanding of such complex mechanisms leading to amplification of chirality is the key to a directed design of such reactions and catalysts. The most prominent example of such an autocatalytic process is the Soai reaction.1 In this presentation mechanistic investigations and a novel mechanism of the Soai reaction will be discussed and strategies to transfer the knowledge to new reactions will be presented.
In particular stereolabile interconverting catalysts open up the possibility of directing enantioselectivity in asymmetric synthesis by formation of diastereomeric complexes with chiral auxiliaries.2,3 The successful realization of such a system by decoration of the ligand backbone with chiral recognition sites attached to a structurally flexible phoshoramidite type catalyst, that can sense the chirality and induce enantioselectivity, is presented.4 Structural flexibility and sensing of the chirality of product molecules result in a rapid increase of enantioselectivity of the dynamic catalysts (Δee of up to 76%) and a shift out of equilibrium.
Literature
1. K. Soai, T. Shibata, H. Morioka, K. Choji, Nature 1995, 378, 767-768.
2. F. Maier, O. Trapp, Angew. Chem. Int. Ed. 2012, 51, 2985-2988.
3. G. Storch, O. Trapp, Angew. Chem. Int. Ed. 2015, 54, 3580-3586.
4. G. Storch, O. Trapp, Nature Chemistry 2017, 9, 179-187.
25 January 2018 – Dr. Jessica Rodríguez-Fernández, Brightlands Materials Center
Nanomaterials with Tailored Optical Properties: from Rational Design to Processing
In this presentation I will give an overview of a wide range of rationally-designed metal- and semiconductor-based nanomaterials with tailored optical properties.[1-3] For instance, it will be shown how colloidal chemistry, cation exchange reactions, and bio-inspired approaches can be deployed to obtain tailor-made nanoparticles and nanostructures with intriguing optical properties. Finally, and with a view toward industrial take up, it will be shown how different inorganic nanoparticles can be successfully processed into industrially-relevant polymer matrixes.
Literature
[1] A.J. Blanch, M. Döblinger, J. Rodríguez-Fernández, Small, 2015, 11, 4550-4559.
[2] M.A.H. Muhammed, M. Döblinger, J. Rodríguez-Fernández, J. Am. Chem. Soc., 2015, 137, 11666–11677.
[3] V. Baumann, M.A.H. Muhammed, A.J. Blanch, P. Dey, J. Rodríguez-Fernández, Isr. J. Chem., 2016, 56, 195–213.
1 February 2018 – Prof. Dr. Tobias Weidner, Aarhus University
A molecular view on protein control of surfaces
Proteins can act as Nature’s engineers at interfaces and manipulate both hard and soft tissue – they can shape biominerals, manipulate cell membranes and control water. Despite the apparent importance for chemists working in the fields of biomineralization, surface engineering and drug delivery the molecular mechanisms behind interfacial protein action have largely remained elusive. We use static and time resolved sum frequency generation spectroscopy combined with computer simulations to determine the structure and the mode of action by which these proteins interact with and manipulate interfaces. Here, I discuss our recent advances in the study of protein driven nucleation
Taking clues from Nature we aim at understanding biomineralization processes at the molecular level to develop design rules for biogenic nanophase materials. Especially the high fidelity control of nanostructured silica within diatoms has been the envy of material scientists for decades. Where diatoms can grow silica using proteins at cell interfaces under ambient conditions, we still need high pH and harsh conditions to structure silica. Despite the apparent importance for physicists and chemists working in the fields of biomineralization, surface engineering, drug delivery, or diagnostics, the molecular mechanisms behind interfacial silica protein action have remained largely elusive. Our goal is to probe the structure and structural dynamics of such active proteins – in action at the surface. As a first step we study the diatom silica peptide R5 when interacting with silica. We use methods based on theoretical and experimental sum frequency generation spectroscopy combined with computer simulations to determine the structure and the mode of action by which these proteins interact with and grow extended 2D silica interfaces.[1]
A particularly fascinating example of protein driven nucleation and phase transitions are ice-nucleating proteins. These proteins are used by specific bacteria to attack plants and cause frost damage by growing ice crystals at temperatures that would otherwise not allow ice formation. A recent survey by the NASA found that biogenic ice nucleators in the troposphere may affect global precipitation patterns. We have followed the interaction of biogenic ice seeding proteins with surrounding water to gain a detailed picture of protein-driven ice nucleation.[2]
Literature
[1] H. Lutz, V. Jaeger, L. Schmüser, M. Bonn, J. Pfaendtner, T. Weidner
The structure of the diatom silaffin peptide R5 within freestanding two-dimensional biosilica sheets Angewandte Chemie Int. Ed., 56, 8277-8280 (2017).
[2] Pandey, R.; Usui, K.; Livingstone, R. A.; Fischer, S. A.; Pfaendtner, J.; Backus, E. H. G.; Nagata, Y.; Fröhlich-Nowoisky, J.; Schmüser, L.; Mauri, S.; Scheel, J. F.; Knopf, D. A.; Pöschl, U.; Bonn, M.; Weidner, T., Ice-nucleating bacteria control the order and dynamics of interfacial water. Science Advances, 2 (2016).
27 April 2017 – Dr Sebastian Kunz, Universität Bremen
Ligands on Nanoparticles – Combining the Benefits of Homogeneous and Heterogeneous Catalysis
A considerable benefit of supported metal nanoparticles (NPs) over homogeneous catalysts is their handling in terms of catalyst recovering and performing continuous processes. However, homogeneous catalysts exhibit significant advantages regarding selectivity control. Especially, chemo- and stereo-selectivity, which are fundamental challenges in heterogeneous catalysis, can be achieved with homogeneous catalysts by application of tailored ligands that guide the reaction through specific interactions with the reactant.
Recently, it has been demonstrated that the use of ligands is not restricted to homogenous catalysis. We demonstrated that chemo- and even stereoselectivity can be achieved with supported NPs when using ligands.[1,2] Thereby the ligands do not act as spectators but they interact with the reactant. This can lead to enhanced activities and allow for the unique molecular control over selectivity known from homogeneous catalysis. We developed a model that describes the origin of stereoselectivity on a molecular level and by application of general guiding principles from asymmetric homogeneous catalysis we were able to raise enantiomeric excesses above 80 %, the limit for industrial relevance.[3] These results highlight the possibility to transfer fundamental principles from homogeneous to heterogeneous catalysis by ligand functionalization and the enormous potential arising from this idea for the development of novel highly active and selective catalysts.
Literature
(1) Schrader, I.; Warneke, J.; Backenköhler, J.; Kunz, S. J. Am. Chem. Soc. 2015, 137, 905.
(2) Schrader, I.; Neumann, S.; Himstedt, R.; Zana, A.; Warneke, J.; Kunz, S. Chem. Commun. 2015, 51, 16221.
(3) Schrader, I.; Neumann, S.; Sulce, A.; Schmidt, F.; Azov, A.; Kunz, S. 2017, under revision.
4 May 2017 – Prof Dr Anton Darhuber, Technische Universiteit Eindhoven
Controlling the conformation of droplets and thin liquid films
This presentation will showcase past and ongoing research efforts concerning the deposition and manipulation of droplets and thin liquid films in the group Mesoscopic Transport Phenomena. I will briefly introduce the underlying physics and highlight several strategies for controlling the shape and distribution of liquids in contact with solid surfaces. Chemical and topographical surface patterning can be employed to impart a desired shape to a droplet, to confine liquid to certain regions on a substrate or to induce an instability and break-up of thin liquid films. While liquids in closed channels can be transported by means of pumps and corresponding pressuregradients, liquid films that maintain a free liquid-air interface can be efficiently mobilized by modulation of the liquid-air or liquid-solid interface energies [1]. Liquids typically flow towards local minima of a non-uniform surface temperature field. If the local temperature T(x,y,t) can be adjusted dynamically, e.g. by means of laser irradiation or integration of micro-heater arrays, then droplets can be shuttled between desired locations on a substrate. Concentration gradients of adsorbed or dissolved chemical components provide another possibility for flow actuation. These allow for higher actuation speeds, because surface tension differences due to composition modulation are usually larger than those due to temperature gradients. I will show that infrared illumination using switchable multimirror arrays can be used for pattern formation in the context of the solution deposition of functional materials and the fabrication of organic electronic devices [2]. Lastly I will discuss ongoing studies regarding flows induced by photochemical reactions.
References
[1] A. A. Darhuber and S. M. Troian, Principles of microfluidic actuation by modulation of surface stresses, Annu. Rev. Fluid Mech. 37, 425 (2005).
[2] J. A. Vieyra Salas, J. M. van der Veen, J. J. Michels, and A. A. Darhuber, Active Control of Evaporative Solution Deposition by Modulated Infrared Illumination, J. Phys. Chem. C 116, 12038 (2012).
18 May 2017 – Dr Christos Markides, Imperial College London
Next-generation high-efficiency solar and waste-heat energy conversion technologies
Solar technologies are projected to deliver the majority of the world’s electricity by 2050, while in the interim concerted efforts will continue to be made so as to utilize fossil fuels as efficiently as possible; in particular, global waste energy in the form of rejected heat to the environment currently accounts for about 250 EJ or 60% of all consumed primary energy. In the case of solar, hybrid PV-thermal (PV/T) collector technology uses a contacting fluid flow that cools the PV cells, thus increasing their electrical efficiency while delivering a useful thermal output, and offers advantages when there is demand for both heat and power, and space is at a premium. Although both solar energy and recovered waste-heat can be used to provide hot water/steam or space heating and electrical power, as well as cooling if required, by far the most common use of the thermal output from PV/T systems and of recovered waste-heat is to use this either directly to provide hot water or steam, or in heat-integration schemes in industrial applications. Nevertheless, a wide range of opportunities arise at higher temperatures, when power-generation or cooling cycles can be employed. These additional options become viable at temperatures typically above ~80 °C, and importantly, become increasingly efficient at progressively higher temperatures. Operating solar panels efficiently at these temperatures is a significant scientific and engineering challenge, since the two modes of heat-transfer loss that reduce their performance, namely convection and radiation, are both exacerbated at higher temperatures, and efforts are being made to alleviated these losses in evacuated-collector designs with optimized surfaces and selective coatings. Similarly, a significant interest exists in improving the technoeconomic performance and value proposition of power-generation systems based on organic Rankine cycles (ORCs) whose current payback times need to be significantly reduced in order to enable the widespread uptake of this technology, including work on the design and use of novel working fluids (including mixtures) and new expander technologies, targeted specifically at the application temperatures and scales of interest. This talk will discuss recent advances in PV/T and ORC technology for high-efficiency conversion of generated or recovered heat to useful power in these applications.
8 June 2017 – Prof Dr Jan-Dierk Grunwaldt, Karlsruher Institut für Technologie
Advanced characterization tools for watching catalysts at work
Probing the structure of catalysts during their preparation, their activation and under reaction conditions is a key for a targeted catalyst design. Furthermore, it is important to understand their dynamic nature, also under varying reaction conditions as occurring in exhaust gas catalysis or energy related processes in the frame of the “German Energiewende”. X-ray techniques such as X-ray absorption spectroscopy (XAS) are valuable tools for this purpose as element-specific information can be received both on amorphous and crystalline materials and, in particular, under reaction conditions while measuring the catalytic activity. Nowadays, it is well known that it is not sufficient to uncover the structure of catalysts before and after a catalytic reaction (ex situ) but such structural information is required during the catalytic reaction (operando). The same is true for materials synthesis. Also in this case in situ studies are indispensible for a deeper understanding.
Since the use of hard X-rays allows penetrating a number of window materials and reaction media, this opens up the design of in situ cells imitating the conditions in conventional reactors. This approach – the structural identification by XAS and simultaneous catalytic experiments – will be illustrated for some example reactions. Even at elevated pressure, it is possible to monitor the structure of heterogeneous catalysts and thus to establish structure-activity relationships. Other opportunities of this technique and related ones are time-resolved studies in the ms-scale, structural identification of low-concentrated constituents of interest (e.g. promoters) and high resolution XAS using high-energy resolved fluorescence detectors. Particular focus has recently been given on new photon in/photon out techniques and X-ray emission spectroscopy.
Finally, both on the macro- and the microscale spatially resolved studies are emerging which are expected to be very important in future. These X-ray microscopic studies combined with XAS, XRD or XRF contrast are strongly complementary to electron microsocopy and using hard X-ray ptychography even a spatial resolution below 10 nm has recently been achieved ex situ and 20 nm in situ.
22 June 2017 – Prof Alexei Lapkin, University of Cambridge
Continuous flow synthesis: from DFT models to process design
Continuous flow processes are seen as a future of manufacturing of speciality chemicals and pharmaceuticals. However, continuous processes could give worse performance that batch processes: this depends on the kinetic scheme, on physical properties of the system and on separation of the products and catalysts. Thus, a mixture of scale-up and process model questions must be answered to develop the most optimal process option. We are developing a generic approach to process design, combining first principles modeling with design of experiments (DoE) driven either by models or by statistics. The ultimate objective of our work is to be able to co-develop optimal reaction system configuration and its validated predictive model in the shortest possible time and smallest number of experiments. Thermodynamic and quantum chemistry calculations provide a significant amount of a priori data about a chemical system. These data can be used to develop initial, not very good, models. Existence of a model allows for model-based DoE to be used and for exploration of different hypotheses about the reaction mechanism, approaches to separation (work-up), recycling schemes, and so on. However, in some cases developing such predictive model will take simply too long. This usually is the case when reaction systems are complex: multi-phase, with very few states being observed. In this case statistical DoE and robotic experimentation offer an alternative approach to process development. A very rapid convergence to satisfactory process recipes was shown in several case studies of reactions being optimised by ‘black-box’ machine learning algorithms. The remaining challenge is to combine the experimental and computational efficiency of machine learning DoE with the desire to develop mechanistic predictive models, to ultimately allow model-based economic control and optimisation of most commercial processes.
6 July 2017 – Prof Esther Amstad, École polytechnique fédérale de Lausanne
Drops: A tool to structure materials
Drops are often used to produce particles of a defined size, shape, and composition. For example, pharmaceutical and food industries often employ airborne drops to produce particles through spray drying. In this case, spray-dried particles are typically spherical and their size scales with that of the drops. Drops can also be used to tune the structure of these particles, which influences their properties. This can be achieved by controlling the evaporation rate of the solvent. If drops are made sufficiently small, such that they have a high surface-to-volume ratio and therefore dry very quickly, they can even produce amorphous particles from materials that have a high propensity to crystallize. In the first part of this talk, I will present a microfluidic spray-dryer that produces µm-sized drops, which are surrounded by fast-flowing air. These drops dry so quickly that crystallization of solutes, contained in the drops, is kinetically suppressed. Hence, the resulting spray-dried particles are amorphous even in the absence of any crystallization inhibiting additives. This is particularly beneficial for the formulation of hydrophobic substances, such as many newly developed drugs, whose bioavailability is limited by their slow dissolution rates and low solubility.
A possibility to produce much larger particles of controlled sizes and compositions is the use of emulsion drops as templates. These drops can be converted into particles that are dispersed in a solution by solidifying their content. The liquid-liquid interfaces of these emulsion drops offer possibilities to tune the surface chemistry of particles that are produced from them. In the second part of the talk, I will show how we use these liquid-liquid interfaces to tune the surface chemistry of particles and capsules, thereby making them responsive to external stimuli or making them mechanically very stable. However, these particles and capsules are only truly useful, if they can be produced in sufficient quantities. In the last part of my talk, I will present a parallelized device that produces monodisperse drops at sufficiently high throughputs to use them as building blocks for macroscopic materials whose structure can be tuned with the size and arrangement of drops.
13 July 2017 – Dr Serhiy Cherevko, Helmholtz Institute Erlangen-Nürnberg
Electrocatalysis in fuel cells and water electrolysers
Almost 90 percent of human carbon dioxide emissions come from fossil fuel combustion, mainly in electricity production and transportation. In both sectors, however, complete carbon neutrality is possible by efficient exploitation of renewable energy sources such as solar and wind. To a high extent this is already realized in electricity generation in Germany. As for transportation, transition to the use of carbon free hydrogen, as the primary means for storing and transporting renewable energy, in electric fuel cell autos is debated. While ecological and social advantages of such transition are obvious, economical concerns are considerable as the initial investments are high, while the operation efficiencies of modern water electrolysers (for hydrogen production) and fuel cells (for on-board electricity generation) are low. In this context, tremendous research activities are targeting at the development and advancement of new superior electrocatalyst materials for both electrochemical technologies.
In the first part of this talk I will give a short introduction into modern hydrogen fuel cells and water electrolysers and challenges they face, main electrochemical reactions, state-of-the-art electrocatalyst materials such as Pt and IrO2 and their limitations, and major experimental techniques in their analysis. The second part of the talk will be devoted to advanced electrocatalyst characterization methods, e.g. involving hyphenated techniques such as scanning flow cell inductively coupled plasma mass spectrometry (SFC-ICP-MS), developed by HI-ERN researchers and our recent experimental results on oxygen and hydrogen electrocatalysis. Here, accent is put on fast high-throughput catalyst libraries screening and stability evaluation, aiming for finding active, stable, and ideally abundant catalysts for especially challenging oxygen evolution and oxygen reduction reactions. Hence, hydrogen electrocatalysis will be covered only briefly.
20 July 2017 – Prof Dr Daniel Klinger, Freie Universität Berlin
From Functional Polymers to Nanomaterials
Nature offers the most sophisticated examples of how selectively tailored polymers, polymer assemblies, and interfaces provide well-defined nanoscopic architectures that result in specific (macroscopic) functions. This outstanding ability of property control via molecular and structural design has long motivated researchers to strive for similar power in synthetic nano-materials. While polymers represent a promising class of materials that allows realizing specific chemical functionalities, dynamic properties and flexibility, simultaneously controlling internal morphology, overall shape, and chemical functionality in a single nanoscopic system remains a challenge. Hence, the ability to realize effective multifunctionality is still in its infancy and unfortunately current examples concentrate on each aspect separately: cooperative dynamic systems are not being fully realized. To address this need, research in the Klinger Lab focuses on the concurrence of chemical functionality and morphological control in polymer colloids and nanomaterials. Following a hierarchical design concept that combines the development of tailor-made macromolecular building blocks with polymeric self-assembly, colloidal chemistry, and interfacial physics, we are developing artificial nanomaterials that will serve as building blocks for advanced technologies such as photonics, drug delivery, catalysis and lithography.
Details of the colloquia which took place from Winter Semester 2009/10 to Winter Semester 2016/17 (Organizer: Prof Dr Thorsten Pöschel ) can be found here.






